Two in Three (65%) Canadians Would Take a Drug Containing Cannabis if Prescribed by a Doctor and Covered by Insurance
Momentum Public Relations
Press Release: February 26, 2019
When It Comes to Cannabis, Trust in Doctors’ Judgement Runs High
- Majority of Canadians (54%) believe taking cannabis for medical reasons without consulting a doctor poses a risk.
- Knowledge of Canadian clinical trials investigating cannabis medicines is low.
- Slim majority of Canadians agree that their doctor is informed enough to treat them with cannabis.
- Majority of Canadians do not consider themselves as cannabis users.
OTTAWA, Feb. 26, 2019 /CNW/ – According to a recent Ipsos poll commissioned by Tetra Bio-Pharma, it seems that Canadians believe that Doctor Knows Best when it comes to recommending cannabis for a medical ailment, even though they are less convinced that they are well enough informed to treat them with it. That concern should change as cannabinoid derived prescription drugs become approved by Health Canada, available in pharmacies and covered by private and/or public drug plans. That time is soon approaching as numerous clinical trials investigating the safety and efficacy of cannabinoid-derived drugs are currently underway.
The study revealed that two in three (65%) Canadians would be willing (36% very/29% somewhat) to take a pharmaceutical drug containing cannabis that their doctor prescribed, if it was approved by Health Canada and covered by either public or private insurance. Those more likely to be willing to take these drugs include men (69%), those aged 18-34 (72%), and residents of Ontario (71%). By contrast, if these drugs were not covered by public or private insurance and patients had to pay out of pocket, four in ten (38%) would still be willing to do so (14% strongly/25% somewhat), particularly among men (44%), those aged 18-34 (51%), and residents of BC (50%).
According to Dr. Guy Chamberland, Chief Executive Officer and CSO of Tetra Bio-Pharma, a global leader in the discovery and development of cannabinoid-derived products, the Ipsos results reinforce current understanding of consumer and physician behaviors. “Patients are open to cannabis as a medical treatment but want their healthcare professional to be in charge. On the flip side, doctors, medical bodies and payors need the safety and efficacy data that they expect from any drug they prescribe. Treatment with cannabis is complex, which is why the pharmaceutical pathway assures precise dosing and consistent formulation.”
Tetra Bio-Pharma undertook the research to gain a better understanding of the attitudes, behaviours and opinions of Canadians on cannabis drugs, including their confidence in taking them, the incidence of medical conditions they have where cannabis treatment could be used, as well as any barriers to cannabis drugs.
While much attention has been paid to the recent legalization of recreational cannabis in Canada, the Ipsos study shows that 69% of respondents do not consider themselves to be cannabis users. Of those that do, half say they use it for recreational purposes. Approximately a quarter of these users report taking it exclusively for medical purposes. If that number seems low, it could be because medical doctors and medical associations are still waiting for scientific data before recommending cannabis to their patients.
New Avenues for Pharmaceutical Research
Canadians seem confident that cannabis has a therapeutic benefit and would be willing to take it for pain and associated symptoms. An overwhelming majority (82%) of Canadians agree (34% strongly agree/48% somewhat) that cannabis can reduce pain and other symptoms. Furthermore, over two-thirds (68%) of Canadians are willing (30% very/38% somewhat) to take cannabis to help manage chronic pain, insomnia, anxiety, or depression. This is potentially interesting for those who suffer from conditions that could be treated with cannabis-based medicines. Not surprisingly, this proportion reaches 84% (60% strongly/24%) among those who suffer from three or more conditions that have been linked to treatment with a cannabis-based medicine.
When it comes to expanding access for patients to cannabis medicines through the regulatory pathway, one in six Canadians (17%) say they are closely following the progress being made with Health Canada approving clinical trials for cannabis-based medicines, while just over four in ten (41%) say they have heard of this development, but are not following it at all. Another 18% said they thought cannabis medicines already existed and the remaining one-quarter (24%) said they were not at all aware.
While cannabis is being authorized for medical purposes under the Cannabis Act, the products currently available are not Health Canada approved prescription drugs. Several clinical trials are underway in Canada with a view to investigating the safety and efficacy of cannabinoid-derived pharmaceuticals. Understandably, those suffering from one or more conditions that could be treated with medical cannabis are more likely to be following this news closely. Furthermore, over four in ten (43%) report being willing (23% very/21% somewhat) to participate in a clinical trial testing cannabis-based medicines if these trials are approved by Health Canada and they were qualified for the trial.
Doctor Knows Best
Canadians seem to recognize that turning to cannabis to manage a health condition is not something they should do on their own. Almost nine in ten (88%) believe that there is some degree of risk (25% major/29% moderate/34% minor) in taking cannabis for health conditions without consulting a physician. This should be a comforting thought considering the side effects that cannabis can have for individuals suffering from mental illness, cardiac conditions and diabetes, among other conditions.
Canadians’ level of trust in their doctor remains high when it comes to their judgement over whether to prescribe cannabis. Just under three-quarters (72%) agree (29% strongly /43% somewhat) that they would trust a drug containing cannabis if their doctor prescribed it. Interestingly though, not everyone is fully confident that their doctor is up-to-date on cannabis treatments. Only a slim majority of Canadians agree that their doctor knows how to treat them with cannabis (56%; 14% strongly/42% somewhat) and that their doctor is sufficiently well-informed on how to do so (53%; 13% strongly/41% somewhat). This suggests that Canadians believe there is room for an improvement in knowledge. It is widely believed that the integration of cannabinoid-derived drugs within the Canadian medical system will become a catalyst for the education and training of doctors on how to prescribe cannabis, something that is complex given the need for precise dosing and consistent product quality.
If ordinary Canadians are open to taking cannabis-based medicines, it seems that doctors are also receptive to prescribing them to patients. While only one in ten (11%) Canadians has asked their doctor to prescribe cannabis for a health condition, 45% of those who did so report that their doctor prescribed cannabis willingly. Another 20% said their doctor prescribed cannabis, but was hesitant, while 11% reported that their doctor prescribed another drug instead.
About the Survey
These are some of the findings of an Ipsos poll conducted between December 7 and December 10, 2018, on behalf of Tetra Bio-Pharma. For this survey, a sample of 2,002 Canadians aged 18+ was interviewed online via the Ipsos I-Say panel. The poll is accurate to within ±2.5 percentage points, 19 times out of 20, had all Canadian adults been polled.
About Tetra Bio-Pharma
Tetra Bio-Pharma (TSX-V: TBP) (OTCQB: TBPMF) is a biopharmaceutical leader in cannabinoid-derived drug discovery and development with a Health Canada approved and FDA reviewed clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. Tetra Bio-Pharma has subsidiaries engaged in the development of an advanced and growing pipeline of Bio Pharmaceuticals and Natural Health containing cannabis and other medicinal plant-based elements. With patients at the core of its mission, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies. For more information visit: www.tetrabiopharma.com.
SOURCE Tetra Bio-Pharma Inc
View original content to download multimedia: http://www.newswire.ca/en/releases/archive/February2019/26/c6186.html
- Published in Life Sciences, Medical Marijuana, News Home, Tetra Bio Pharma
Sirona Biochem to Exhibit at PCHi 2019 in Guanzhou China
Momentum Public Relations
Press Release: February 26, 2019
Sirona Biochem Corp. (TSX-V: SBM) (FSE: ZSB) (the “Company“) announces that it will exhibit at The Personal Care and Homecare Ingredients trade show (PCHi 2019) in Guanzhou China Feb 26-28th 2019. The PCHi 2019 trade show is China’s #1 sourcing platform for the global personal care industry https://www.pchi-china.com/en/. Sirona will be principally promoting its novel skin lightening agent TFC-1067 in addition to its anti-wrinkle and anti-aging compounds.
Sirona’s business development team in China, will be supported by a large Chinese cosmetic distribution company. The distribution company is working closely with Sirona through a memorandum of understanding to establish an optimal pathway to commercialize TFC-1067 into the Chinese skin lightening market. The PCHi 2019 will introduce Sirona’s technology to an estimated 75 cosmetic companies and numerous market experts. The business development team assembled has local knowledge and expertise that allows Sirona to navigate this immense opportunity.
Thanks to the efforts of PRC Partners Limited, Sirona has established a strong and experienced team with the diverse skill to navigate the optimal pathways to commercialize into this market. The global skin lightening market is estimated to grow to US$31.2 billion by 20241. Asia makes up the largest and fastest growing segment of the market.
Sirona would also like to thank the strong support for our recently announced financing which has been filed with the TSX Venture exchange. The Offering is subject to TSX Venture Exchange (“TSXV“) final acceptance of requisite regulatory filings. When approved by the TSXV, a news release will be issued by the company.
About Sirona Biochem Corp.
Sirona Biochem is a cosmetic ingredient and drug discovery company with a proprietary platform technology. Sirona specializes in stabilizing carbohydrate molecules with the goal of improving efficacy and safety. New compounds are patented for maximum revenue potential.
Sirona’s compounds are licensed to leading companies around the world in return for licensing fees, milestone fees and ongoing royalty payments. Sirona’s laboratory, TFChem, is in France and is the recipient of multiple French national scientific awards and European Union and French government grants. For more information, please visit www.sironabiochem.com.
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
- Published in Business, Life Sciences, News Home, Sirona Biochem
Crystal Lake gets approval for option agreement for Newmont Lake project
Momentum Public Relations
Press Release: February 25, 2019
The TSX Venture Exchange has accepted for filing a mineral property option agreement dated for reference Nov. 29, 2018, between Crystal Lake Mining Corp. and Romios Gold Resources Inc. The option agreement was approved on February 22, 2019. Under the agreement, Romios has agreed to grant Crystal Lake an option to acquire up to a 100-per-cent interest in the Newmont Lake project located in British Columbia.
In order to exercise the option Crystal Lake must:
- Complete $8-million in exploration expenditures on the Newmont Lake project over the next three years ($3-million in the first year and $2.5-million in each the second and third years);
- Pay $2-million in cash to Romios as follows: $250,000 on signing of agreement (paid) and a further $250,000 payable on each of 90 days, 180 days and 270 days following exchange approval of the transaction, with a final $1-million payment upon Crystal Lake earning its 100-per-cent interest in the Newmont Lake project;
- Issue a total of 12 million common shares of Crystal Lake to Romios over a three-year period as to four million each year, with the first four million shares to be issued following exchange approval of the transaction. The Company is issuing the first tranche of 4,000,000 shares to Romios having a legend of a six month hold period on the first 1,000,000 shares and a twelve month hold period on the balance of 3,000,000 shares.
Romios will retain a 2-per-cent net smelter return royalty on the Newmont Lake project, or on any after acquired claims within a five-kilometre radius of the current boundary of the project, which may be reduced at any time to a 1-per-cent NSR on the payment of $2-million by Crystal Lake per 0.5 per cent of the NSR. Crystal Lake will issue two million shares to Romios in the event that a National Instrument 43-101 technical report on the Newmont Lake project contains a resource estimate which exceeds one million ounces of gold equivalent resources (being the sum of indicated and inferred). Crystal Lake will issue an additional one million shares to Romios for each full one million additional ounces of gold equivalent resources so documented in a National Instrument 43-101 technical report on the Newmont Lake project.
About the Company
Crystal Lake Mining Corporation is a mineral exploration/Development company focused on creating value through the exploration and development of its British Columbia and Ontario mineral properties.
We seek Safe Harbor.
- Published in Crystal Lake Mining, Mining, News Home
Canada Cobalt Works Scheduled to Present in the Corporate Presentation Forum at PDAC 2019
Momentum Public Relations
Press Release: February 24, 2019
Canada Cobalt Works Inc. (TSXV: CCW) (OTC: CCWOF) (Frankfurt: 4T9B) (the “Company” or “Canada Cobalt”) is pleased to announce that President and CEO Frank J. Basa is scheduled to make a presentation to investors as part of the Corporate Presentation Forum at PDAC 2019 in Toronto.
The presentation will be in the Energy Metals and Specialty Products session on Tuesday March 5 at 11:15 am Eastern in Room 803 in the South Building of the Metro Toronto Convention Centre.
Mr. Basa’s presentation will focus on Canada Cobalt’s recent high-grade drill results, a recent apparent gold system discovery, its plans to bring the cobalt-silver Castle Mine back into production and the technological lead that the Company has achieved with its environmentally green Re-2OX hydrometallurgical process, which has been demonstrated in lab tests to produce cobalt sulphate to the specifications required by electric battery manufacturers without the need for smelting.
Mr. Basa will be available for questions after the presentation and also will meet with investors during PDAC at Booth 2738 in the Investors Exchange exhibit area on the 800 level in the South Building.
Canada Cobalt Works was recently ranked by the TSX among the Top 5 Mining Companies in its 2019 Venture 50 listing of top-performing companies on the TSX Venture Exchange. The Company regards this ranking as a validation of the strategy it has pursued towards building a vertically integrated mining company – from mining and milling through to the production of cobalt and other sulphates that meet the electric-car manufacturers’ requirements for conflict-free cobalt they want for their electric-car batteries.
About Canada Cobalt Works Inc.
Canada Cobalt is a pure play cobalt company focused on its past producing Castle mine in the Northern Ontario Cobalt Camp, Canada’s most prolific cobalt district. With underground access at Castle, a recently installed pilot plant to produce cobalt-rich gravity concentrates on site, and a proprietary hydrometallurgical process known as Re-2OX for the creation of technical grade cobalt sulphate as well as nickel-manganese-cobalt (NMC) formulations, Canada Cobalt is strategically positioned to become a vertically integrated North American leader in cobalt extraction and recovery. More information is available at www.canadacobaltworks.com.
“Frank J. Basa”
Frank J. Basa, P. Eng.
President and Chief Executive Officer
Neither the TSX Venture Exchange nor its Regulation Service Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release. This news release may contain forward-looking statements including but not limited to comments regarding the timing and content of upcoming work programs, geological interpretations, receipt of property titles, potential mineral recovery processes, etc. Forward-looking statements address future events and conditions and therefore, involve inherent risks and uncertainties. Actual results may differ materially from those currently anticipated in such statements.
SOURCE Canada Cobalt Works Inc.
View original content: http://www.newswire.ca/en/releases/archive/February2019/25/c1830.html
Contact:
Frank J. Basa, P.Eng., President and CEO, 1-819-797-4144, or Wayne Cheveldayoff, Corporate Communications, waynecheveldayoff@gmail.com, 1-416-710-2410
- Published in Canada Cobalt Works, Mining, News Home
Grown Rogue Announces Binding Agreement for Michigan Cannabis Licenses and Assets
Momentum Public Relations
Press Release: February 25, 2019
Grown Rogue International Inc. (CSE:GRIN | OTC: NVSIF) (“Grown Rogue” or the “Company“), a vertically-integrated, multi-state cannabis company, with licenses and operations in Oregon and California, has signed a binding agreement which provides the Company the right to acquire operational control of certain cannabis licenses and related assets as part of its expansion into the Michigan cannabis market, pending certain regulatory approvals.
“With the second highest total number of medical cannabis card holders in the United StatesMichigan’slegalization of cannabis for adult-use presents a very large cannabis market opportunity. Significant barriers to entry at the local level add meaningful value to the limited number of municipal licenses approved,” explained Obie Strickler, CEO of Grown Rogue. “We are very diligent in our expansion strategy and in finding the right partners to take the proven Grown Rogue platform into the state. Combining our expertise enables us to efficiently navigate and comply with the regional regulatory environment and rapidly expand the Grown Rogue footprint and brand.”
Grown Rogue has entered into a binding agreement (the “Agreement“) with Blue Zebra Community LLC (“Blue Zebra“). The Agreement provides for the option to acquire operational control (the “Option“) of the following cannabis related assets in Michigan pending Municipal and State regulatory approval (the “Michigan Assets“):
- Two strategically located proposed provisioning centers (retail dispensaries) in high demand regions in Midtown Detroit and Hazel Park where limited municipal licenses have been granted.
- A proposed 19,000 sq ft indoor cultivation and processing facility in Detroit, Michigan capable of producing 1,500,000 grams of dried cannabis flower annually at full production; and
- An entity that has received multiple municipal cultivation licenses for a 28-acre parcel located in the northern portion of the lower Michigan peninsula.
Once fully licensed, the provisioning (retail) centers will offer multiple Michigan cannabis brands as well as locally produced Grown Rogue branded products for the Michigan cannabis market. This will launch the Grown Rogue brand into the Midwest region.
Upon exercising the Option by Grown Rogue, Blue Zebra (or affiliates) is required to assign its rights and obligations to the Company pursuant to a binding agreement Blue Zebra (or affiliates) has with Helios Holdings, LLC (“Helios“) which provides the framework for the acquisition of the Michigan Assets as described below (the “Helios Agreement“). As part of its Agreement with Blue Zebra, Grown Rogue will issue 2,212,876 common share purchase warrants to Blue Zebra (or its affiliates) with an exercise price of $0.44 per share (the “Warrants“), which vest according to certain milestones in accordance with the agreement with Blue Zebra.
The Warrants expire on June 20, 2023. Grown Rogue will have the right to accelerate the expiry date of 25% of the Warrants during the term if the shares of the Company close at or above $1.00 per share for a period of twenty (20) consecutive days. An additional 25% of the Warrants will accelerate if the shares of the Company close at or above $1.50 per share for a period of twenty (20) consecutive days, and the remainder of the Warrants will accelerate if the shares of the Company close at or above $2.00 per share for a period of twenty (20) consecutive days.
Pursuant to the terms of the Agreement, Grown Rogue has granted Blue Zebra, together with any affiliates, a pre-emptive right to maintain ownership, should the Warrants be exercised, of up to 5% of Grown Rogue’s common shares. At the time Blue Zebra, or any affiliates, exercises its Warrants to obtain 5% ownership in the Company, Blue Zebra will have the right to nominate one member to Grown Rogue’s board who shall be nominated by management at each annual shareholder meeting of Grown Rogue until such time the Blue Zebra’s ownership in Grown Rogue falls below 4.67%. In addition, Grown Rogue has agreed to pay Blue Zebra between 5% and 7% of top line future revenues generated from its licensed operations in Michigan. Payment on these revenues shall be in a combination of stock and cash.
Helios Agreement
Helios, and its affiliate, intend to contribute real property for a 19,000 sq ft. proposed cultivation and processing facility to a newly formed joint venture to be operated by Grown Rogue, or its designated affiliate, upon receiving all necessary regulatory approvals.
Grown Rogue intends to secure a non-dilutive, real estate financing facility to cause the improvements to the Michigan Assets to be completed.
Helios will contribute the remaining Michigan Assets into one or more newly formed operating company(s) (“OpCo“). It is intended that Grown Rogue will hold a 3.62% convertible debenture for the initial funding of OpCo, with a put/call option to acquire all the issued and outstanding shares of the OpCo. The convertible debenture principal, including any interest, shall be returned to Grown Rogue first from 50% of any cash flow generated by OpCo. Conversion of the debenture by Grown Rogue is subject to State and Municipal regulatory approval.
The State licensing and regulatory process in Michigan requires multiple tiers of approval for any operations (cultivation, retail). Several municipal licenses have successfully been awarded to Helios and its affiliates.
Once licensed by the State, the 19,000 square foot facility will be the first cultivation and manufacturing center for OpCo. Initial plans for this facility will include the ability to produce 1,500,000 grams of cannabis flower per year with the construction of a perpetual harvest facility expected to open in Q4 2019. This facility will also include best-in-class extraction facilities where the OpCo will produce branded derivative products.
The interest in the 28-acre cultivation facility can include either indoor or greenhouse operations which are currently being evaluated. Construction and operation of this facility is expected in 2019 with an anticipated yearly yield of between 1,500,000 and 2,500,000 grams of annual cannabis production based on final design plans.
Both of OpCo’s proposed retail dispensaries are located in desirable high traffic locations and are expected to be licensed and operational by Q4 2019. OpCo aims to further expand to 10 retail dispensaries and 50,000 sq ft of cultivation facilities by the middle of 2020.
“Our partnership with Grown Rogue has accelerated our operations and already we have identified several additional licenses which could make Grown Rogue one of the leading cannabis operations in the entire state of Michigan,” stated Maxim Ermakov, Helios Executive Director.
About Grown Rogue
Grown Rogue International (CSE: GRIN | OTC: NVSIF) is a vertically-integrated, multi-state cannabis company curating innovative products to provide consumers with the right cannabis experience. Each of Grown Rogue’s products and strains are categorized and marketed based on unique effects and designed for the full range of a consumers’ lifestyle. Grown Rogue is scaling the vertically integrated model into multiple states by incorporating best-in-class manufacturing facilities and a proprietary distribution platform based on Microsoft technology. Grown Rogue’s diverse cannabis product suite includes premium flower, patent-pending nitrogen sealed pre-rolls, oil and concentrates, and edibles featuring a partnership with world-renowned chocolatier, Jeff Shepherd.
Subscribe to Grown Rogue investor news alerts.
- Published in Cannabis, Grown Rogue, Marijuana, Medical Marijuana, News Home
Canada Cobalt Drills into Bonanza Grades at Castle
Momentum Public Relations
Press Release: February 20, 2019
Canada Cobalt Works Inc. (TSXV: CCW) (OTC: CCWOF) (Frankfurt: 4T9B) (the “Company” or “Canada Cobalt”) is pleased to announce that first-pass underground Phase 1 drilling on the first level of the Castle mine near Gowganda, 75 kilometers from Kirkland Lake, has returned multiple high-grade cobalt and silver intercepts at very shallow depths.
Drill results have also revealed areas overlooked by historical explorers that show potential to host very high-grade “shoots” of silver and cobalt-silver mineralization, mixed with occasional nickel and gold, that may extend to considerable depths outside of historical drilling or workings.
Highlights:
- New discovery of very high-grade silver vein structures approximately 55 meters southwest of the #3 Shaft where a silver discovery in 1979 put the Castle mine back into production for a decade – CA-18-54 cut 93.7 ounces per ton Ag (3,213 grams per tonne) over one meter including 286.3 ounces per ton (9,816 g/t) over 0.33 meters starting just 9.71 meters downhole, with the hole drilled across the structure at 25o to core axis and then bottoming in high-grade mineralization from 18.84 meters to 20.50 meters;
- 385.2ounces per ton silver (13,208 g/t), 0.67% cobalt and 3.77 g/t gold over half a meter within a broader 5.51-meter zone that also included 1.87% cobalt over 2.54 meters and 76.4 ounces per ton Ag (2,620 g/t) over a core length of 5.51 meters starting at just 1.46 meters (CA-18-02, collared near the adit entrance, was drilled perpendicular to the strike of the targeted vein structure, sub-parallel to the dip of the vein);
- All 47 assayed shallow underground test holes intersected cobalt mineralization with an impressive one-quarter of those holes returning high-grade intercepts of 1.05% to 3.7% cobalt over an average core length of 1.77 meters (true widths unknown at this time);
- A series of key initiatives, already in motion, will drive an expanded Phase 2 program to immediately build on these results and accelerate the broader 2019 corporate goal to leverage the company’s proprietary Re-2OX technology simultaneously with heightened activity at the Castle mine in ways that continue to keep CCW share dilution to a minimum.
Jacques Monette, Canada Cobalt Director and career miner, commented: “These results from our first-ever underground drilling support the interpretation that much was left behind at the Castle mine, not just cobalt but very high-grade silver. This array of high-grade intercepts at shallow levels further energizes our team as we ramp up again and implement a greatly expanded second phase of our underground strategy.
“2018 was a pivotal year in the revitalization of the Castle mine,” Monette continued, “and we look forward to achieving key new milestones in the coming months. At the height of winter, we have a very active site and another drill program kicking off this week southeast of the mine as we build on a potential high-grade gold discovery at Castle East. This is an exciting new development as the Archean rocks east of the high-grade silver mines were never tested historically.”
Next Action Steps Underground
- Immediate priority is follow-up drilling targeting high-grade “shoots” near #3 Shaft and adit entrance;
- Through discussions with Canada Cobalt’s engineering consultant, and the Northern Ontario Ministry of Mines and Development, all material from the first level stopes will be dumped and removed in accordance with an amended advanced exploration permit (notably, Canada Cobalt can produce gravity concentrate on site). The stopes will then be available for back-filling with cemented tailings as part of a major new program involving Re-2OX that Canada Cobalt will be elaborating on in the very near future;
- As part of the amended exploration permit, blasting will be undertaken throughout the first level;
- Hydrogeological studies will evaluate dewatering of the remaining 10 levels of the Castle mine.
Underground Drilling Highlight Details
#3 Shaft Area
Drill hole CA-18-54 hit two very high-grade silver intercepts starting just 9.71 meters downhole, 93.7 opt(3,213 g/t) over one meter including 286.3opt (9,816 g/t) over 0.33 meters, followed by 11.8opt(406.1 g/t) over 1.66 meters at the bottom of the hole beginning at 18.84 meters. Drilling was across these structures, with the first intercept at 25o to core axis and the latter intercept cutting a new vein at a lower angle than the first intercept. This is a new discovery where there was limited previous work – follow-up drilling will track the vein, and potential associated veins, to depth.
Adit Entrance Discovery
A further review of historical work in addition to Canada Cobalt drilling including updates of previously released preliminary data, suggests a silver-cobalt rich vein intersected near the adit entrance in the first three drill holes may also extend much further to depth. CA-18-02 was one of several holes that attempted to follow the vein from a series of inclinations from approximately the same drill set-up through the Nipissing Diabase toward the second level in order to test grade potential. Veining is irregular, so contacts averaged approximately 25 degrees to core axis (refer to Nov. 2, 2018 news release).
High-Grade Cobalt Values Confirmed
For the first time at the Castle mine, high-grade cobalt values, along with occasional nickel and gold, have been confirmed through drilling to exist in vein structures, and as much as 174 meters apart (collar to collar) from near the adit entrance (23 meters below shaft collar) to the #3 Shaft area.
Multiple Target Areas Throughout Expansive First Level
Phase 1 drilling has provided the Canada Cobalt geological team with multiple highly prospective new targets for detailed follow-up utilizing a more versatile and powerful drill rig that can track high-grade veins to depth. Many vein structures have yet to be tested. Based on historical mining data, veins at Castle are known to pinch and swell and move in and out of high-grade and lower grade mineralization over considerable distances. The first level (there are 11 in total) is in excellent condition with a footprint of 365 meters east-west and 360 meters north south. A total of 58 holes were completed (47 have been assayed) in Phase 1, totaling 675 meters. A table of assay highlights/details will be posted on the Canada Cobalt web site.
Canada Cobalt Adds $500,000 From Exercise of Warrants
Canada Cobalt has received approximately $500,000 from the exercise of warrants over the past two months and will continue its Northern Ontario Cobalt Camp leading practice of keeping share dilution to a minimum while aggressively advancing its exploration and development strategies in a cost-effective manner.
Quality Assurance/Quality Control
Canada Cobalt Works’ AQTK drill program employs diligent standards in drill core sampling and quality assurance/quality control. Core from the above holes was sent to Swastika Laboratories in Swastika, Ont., for analysis. Where silver was visually and significantly present, a pulp-metallic analysis was completed for the silver assays. The entire sample was dried, weighed and crushed over 95 per cent, then fully pulverized and passed through 147-micron screen to create a plus-147-micron fraction (metallics) and a minus-147-micron fraction (pulp). Approximately 50 grams of the metallic fraction was weighed, fused and assayed by fire assay. The resulting dore bead of gold and silver was weighed before adding HNO3 acid to dissolve the silver. The remaining gold bead was weighed and subtracted from the gold plus silver bead to give a silver weight and grade for the metallics portion. The minus-147 fraction (pulp) had two samples from entire pulp which were weighed and then digested by two-acid aqua regia and finalized by AAS reading for cobalt, silver and nickel. Final silver grade is a calculated weighted average using grades and weights from both pulps and metallics.
As gold was not assayed in the pulps, the final gold assay was calculated by weighted average, as for the silver, but assuming a grade of zero gram per tonne gold for the pulp portion. Cobalt and nickel were reported as a percentage grade. Other samples were assayed for cobalt, silver and nickel by AAS after aqua regia digestion. Analytical accuracy and precision are monitored by the analysis of reagent blanks and reference materials at the lab. Quality control is further assured by the insertion of blind certified standard reference material and blanks into the sample stream at regular intervals by Canada Cobalt Works personnel in order to independently assess analytical accuracy.
Qualified Person
The technical information in this news release was prepared under the supervision of Frank J. Basa, P.Eng., Canada Cobalt’s President and Chief Executive Officer, who is a member of Professional Engineers Ontario and a qualified person in accordance with National Instrument 43-101.
About Canada Cobalt Works Inc.
Canada Cobalt is a pure play cobalt company focused on its past producing Castle mine in the Northern Ontario Cobalt Camp, Canada’s most prolific cobalt district. With underground access at Castle, a recently installed pilot plant to produce cobalt-rich gravity concentrates on site, and a proprietary hydrometallurgical process known as Re-2OX for the creation of technical grade cobalt sulphate as well as nickel-manganese-cobalt (NMC) formulations, Canada Cobalt is strategically positioned to become a vertically integrated North American leader in cobalt extraction and recovery.
“Frank J. Basa”
Frank J. Basa, P. Eng.
President and Chief Executive Officer
Neither the TSX Venture Exchange nor its Regulation Service Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release. This news release may contain forward-looking statements including but not limited to comments regarding the timing and content of upcoming work programs, geological interpretations, receipt of property titles, potential mineral recovery processes, etc. Forward-looking statements address future events and conditions and therefore, involve inherent risks and uncertainties. Actual results may differ materially from those currently anticipated in such statements.
SOURCE Canada Cobalt Works Inc.
View original content: http://www.newswire.ca/en/releases/archive/February2019/19/c9297.html
Contact:
Frank J. Basa, P.Eng., President and CEO, 1-819-797-4144; or Wayne Cheveldayoff, Corporate Communications, waynecheveldayoff@gmail.com, 1-416-710-2410
- Published in Canada Cobalt Works, Mining, News Home
The Nelligan Project: a Gold Vessel Sailing on a Vast Gold Hydrothermal System
Momentum Public Relations
Press Release: February 19, 2019
Management of Vanstar Mining Resources Inc. (« Vanstar ») provides an update on its Nelligan gold project as well as a retrospective of the drill results highlights from the main gold showings of the Nelligan project.
IAMGOLD Corporation, Vanstar’s partner in the development of the Nelligan project, started last January a new 15,000-meter winter drilling program. 4,750 metres of drilling has been completed so far and the 16 drill holes have intersected their intended targets. The drill cores are being prepared for assays and results will be announced as soon as they become available.
The objectives of the 2019 drilling program is to: 1) refine an initial gold deposit model by merging previous results with current ones 2) support an initial 43-101 resource estimate and 3) help position future drilling campaigns.
«We are very pleased with the progress and quality of the work executed by our partner. The Nelligan project requires patience, efforts and work in order to become a major gold project in Canada. The targets are at the right place and we remain very optimistic that those results will be as good as the previous ones. » stated Mr. Guy Morissette, CEO of Vanstar.
The Nelligan project
The Nelligan project comprises 158 mining cells covering an area of 8,216 hectares located south of Chapais, in Northern Quebec. It includes many important gold zones namely the Renard, 36, Liam, Dan and Lac d’Eu zones. The Liam and Dan zones as well as the east portion of the 36 zone were discovered by drilling in 2013-2014 while the Renard and 36 west zones, located some hundreds of meters north, were found in 2016. The historical showing of the Lac d’Eu, located north-east of the mentioned zones, is known since the mid-1990s.
The Renard zone is located within a vast gold hydrothermal environment measuring 150 to 250 metres in width. This showing was drill intersected over a strike of more than 1 kilometre and at vertical depth of more than 450 metres and appears to be associated with a long corridor corresponding to a low mag anomaly crossing the property from east to west. Drill holes NE-17-61 and NE-17-64 located from 700 metres to 1.2 kilometres west of the Renard zone, both intersected this same horizon with significant gold results (See Sept 5, 2017 Press release). Furthermore, 4 kilometres west of the Renard showing, drill holes done by Bold Ventures in 2017 also intersected gold values ( See March 9, 2017 Press release) thus showing the presence of gold along this corridor.
The Renard zone remains open laterally and at depth.
The 36 zone is located between the Renard and Liam zones and comprises two blocks (east and west) within a silicified, brecciated and hematized geological environment.
The structure of this showing was intersected over many hundreds of metres by 5 to 10 metres wide, at vertical depth of 250 metres. Zone 36 remains open in all directions.
The Liam and Dan zones were drill intersected on widths up to 30 metres, over 700 metres in length and at vertical depth of more than 300 metres. Associated with silicified zones and silicified and hematized conglomerates, the Liam and Dan showings are located near major faults oriented SSW.
All these zones remain open laterally and at depth.
See maps attached
The following table shows highlights from these gold zones.
| Renard Zones | |||
| Drill holes | Width | Grade | |
| (m) | (g/t Au) | ||
| NE-16-44 | 10.34 | 4.43 | |
| NE-16-48 | 11.60 | 3.19 | |
| NE-16-49 | 37.50 | 3.60 | |
| NE-17-64 | 11.28 | 2.78 | |
| NE-18-69 | 30.80 | 2.66 | |
| Including | 15.16 | 3.28 | |
| NE-18-75 | 23.10 | 2.59 | |
| NE-18-84 | 82.60 | 3.31 | |
| NE-18-87 | 27.20 | 3.48 | |
| NE-18-91 | 12.70 | 3.98 | |
| NE-18-95 | 38.16 | 3.59 | |
| Including | 2.60 | 12.80 | |
| NE-18-96 | 24.03 | 5.69 | |
| Including | 5.89 | 19.89 | |
| NE-18-97 | 8.60 | 4.16 | |
| NE-18-98 | 12.12 | 5.04 | |
| Including | 3.46 | 9.79 | |
| NE-18-99 | 13.19 | 2.42 | |
| And | 30.95 | 2.35 | |
| Liam, Dan and 36 zones | |||
| Drill holes | Width | Grade | |
| (m) | (g/t Au) | ||
| NE-13-04 | 20.70 | 2.04 | |
| Including | 10.50 | 2.30 | |
| NE-13-08 | 15.10 | 3.74 | |
| Including | 11.60 | 4.69 | |
| NE-14-17A | 8.40 | 3.04 | |
| NE-14-21 | 19.60 | 2.34 | |
| Including | 4.30 | 5.53 | |
| NE-14-22 | 20.00 | 2.28 | |
| Including | 6.00 | 5.90 | |
| NE-15-25 | 6.00 | 9.58 | |
| NE-16-32 | 12.00 | 3.18 | |
| NE-16-36 | 17.87 | 3.20 | |
| Including | 4.20 | 6.78 | |
| NE-18-81 | 12.20 | 3.93 | |
| NE-18-89 | 14.30 | 4.26 | |
Visible gold and molybdenum were often observed within these gold zones.
IMG- VSR Agreement
According to the 2018 amended agreement, IAMGOLD Corporation can acquire a 75 % interest in the Nelligan project in exchange for providing a 43-101 resource estimate and cash payments for a total of $2,750,000 comprising of 3 annual payments of $400,000 and a residual payment of $1,550,000 on or before 2022. IMG can also acquire an additional 5% interest by delivering a feasibility study. Vanstar would then own a non-contributary 20 % undivided interest (Net Carried Interest), despite the sums to be invested in an eventual production phase.
When a production decision is achieved, IAMGOLD Corporation (IMG) would pay for all the expenses required to complete this ultimate phase. Although, Vanstar would have to participate, prorated to its auto-generated revenues from the Nelligan project, to the reimbursement of the portion of the production phase cost relative to its undivided 20 % interest in the project. In addition, Vanstar owns a 1% NSR royalty on some mining cells that are part of the main gold showings. This royalty was acquired from the original owners.
Also, if Vanstar would sell its 20 % undivided interest to IMG in exchange for an offer based on 2 independent evaluations, Vanstar would obtain, in addition of the sale proceeds of its 20% interest, a 1.5 % NSR royalty on the whole property and a 2.5% NSR royalty for the portion from the original owners.
Vanstar has also received from its partner IAMGOLD a cash payment of $400,000 earlier this month, as part of the 2018 agreement. One officer and a director exercised part of their stock options for proceeds of $18,500 for Vanstar and 950,000 warrants were exercised in 2019 for proceeds of $95,000. The company has more than $2,500,000 in cash.
As part of its operations, the company continues to evaluate different projects and opportunities to improve shareholder value.
This press release was read and approved by Gilles Laverdière, Vanstar’s Geologist and Qualified Person under NI-43-101.
Forward Looking Statement
This news release contains forward-looking statements. All statements, other than of historical fact, that address activities, events or developments that the Company believes, expects or anticipates will or may occur in the future (including, without limitation, statements regarding expected, estimated or planned gold production, cash costs, margin expansion, capital expenditures and exploration expenditures and statements regarding the estimation of mineral resources, exploration results, potential mineralization, potential mineral resources and mineral reserves) are forward-looking statements. Forward-looking statements are generally identifiable by use of the words “will”, “should”, “continue”, “expect”, “anticipate”, “estimate”, “believe”, “intend”, “to earn”, “to have’, “plan” or “project” or the negative of these words or other variations on these words or comparable terminology. Forward-looking statements are subject to a number of risks and uncertainties, many of which are beyond the Company’s ability to control or predict, that may cause the actual results of the Company to differ materially from those discussed in the forward-looking statements. Factors that could cause actual results or events to differ materially from current expectations include, among other things, without limitation, failure to meet expected, estimated or planned gold production, cash costs, margin expansion, capital expenditures and exploration expenditures and failure to establish estimated mineral resources, the possibility that future exploration results will not be consistent with the Company’s expectations, changes in world gold markets and other risks. Any forward-looking statement speaks only as of the date on which it is made and, except as may be required by applicable securities laws, the Company disclaims any intent or obligation to update any forward-looking statement.
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Source :
Guy Morissette
CEO Vanstar Mining Resources Inc.
gmvanstar@gmail.com
819-763-5096
Maps associated with this press release are available at the following addresses:
http://www.globenewswire.com/NewsRoom/AttachmentNg/029f609f-eb90-4edc-8d99-ca09f7a1d62b
http://resource.globenewswire.com/Resource/Download/e4075c68-822f-4273-92fc-95815dc5117c
http://resource.globenewswire.com/Resource/Download/d41a8bed-6af3-4592-8736-418f04a3e207
- Published in Mining, News Home, Vanstar Mining
Tetra Bio-Pharma unit launches Hemp Energy Drink
Momentum Public Relations
Press Release: February 19, 2019
Tetra Natural Health, a subsidiary of Tetra Bio-Pharma Inc., has made its Hemp Energy Drink available in 59 grocery stores and convenience stores in British Columbia, Alberta and Saskatchewan.
Hemp Energy Drink tasting in Kelowna on Feb. 3, 2019
In British Columbia the Hemp Energy Drink is available to consumers in Vancouver, Balfour, Burnaby, Castlegar, Clearwater, Grand Forks, Hope, Houston, Kaslo, Keremeos, Ladner, Langley, Lillooet, Nanaimo, Nelson, North Vancouver, Oliver, Osoyoos, Port Alberni, Robson, Rossland, Salt Spring Island, Simon Fraser University, Slocan Park, Squamish, Summerland, Surrey, Trail, Vernon, Victoria, West Kelowna, Whistler and White Rock.
In Alberta, the Hemp Energy Drink can be found in Athabasca, Banff, Boyle and Langdon, and in Saskatchewan in Kindersley and Tisdale.
The tastings of the Hemp Energy Drink were very popular on Feb. 2 and Feb. 3, during the Wellness Show Vancouver 2019 and at a product presentation in Kelowna. The distribution plan is expected to accelerate the penetration of HED attractive green cans in stores in the coming weeks.
“Our distribution plan is on schedule and the arrival of the Hemp Energy Drink is creating a buzz among consumers as soon as the product becomes available in store,” said Richard Giguere, chief executive officer, Tetra Natural Health. “We will accelerate the deployment of our market opening strategy in a few weeks with the arrival of 1.7 million cans of Hemp Energy Drink, of which more than 600,000 will be quickly shipped to new markets in Canada.”
Hemp Energy Drink is the first energy drink made from hemp in the country. Light, energizing, fresh, Hemp Energy Drink is licensed by Health Canada and contains fewer calories and more natural ingredients than any other energy drink. The product will soon be available in three flavours, classic, mango and raspberry, and it will be accessible in stores in a few months in all regions across Canada.
About Tetra Bio-Pharma Inc.
Tetra Bio-Pharma is a biopharmaceutical leader in cannabinoid-based drug discovery and development with a Health Canada-approved, and Food and Drug Administration-reviewed, clinical program aimed at bringing novel prescription drugs and treatments to patients and their health care providers. The company has several subsidiaries engaged in the development of an advanced and growing pipeline of biopharmaceuticals, natural health and veterinary products containing cannabis and other medicinal plant-based elements. With patients at the core of what it does, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies.
- Published in Business, hemp, Marijuana, Medical Marijuana, News Home, Tetra Bio Pharma
Bluestone Announces $10,000,000 Bought Deal Financing
Momentum Public Relations
Press Release: February 19, 2019
Bluestone Resources Inc. (TSXV:BSR | OTCQB:BBSRF) (the “Company” or “Bluestone”) announces that the Company has entered into an agreement with Cormark Securities Inc. to act as lead underwriter (the “Lead Underwriter”) and a syndicate of underwriters (collectively with the Lead Underwriter, the “Underwriters”) to purchase on a “bought deal” private placement basis (the “Offering”), up to $10,000,000 of units of the Company (the “Units”) at a price of $1.25 per Unit (the “Offering Price”).
Each Unit will consist of one common share of the Company (a “Unit Share”) and one-half of one common share purchase warrant (each whole common share purchase warrant, a “Warrant”). Each Warrant will entitle the holder to acquire one common share of the Company for 24 months from the closing of the Offering at a price of $1.65.
The Company has also granted to the Underwriters the option to sell up to an additional $5,000,000 of Units, exercisable until the date which is two days prior to the closing date.
The net proceeds of the Offering will be used towards advancing the Cerro Blanco Gold Project and for general corporate purposes.
Closing of the offering is anticipated to occur on or before March 12, 2019 and is subject to receipt of applicable regulatory approvals including approval of the TSXV.
This news release does not constitute an offer of securities for sale in the United States. The securities being offered have not been, nor will they be, registered under the Unites States Securities Act of 1933, as amended, and such securities may not be offered or sold within the United States absent U.S. registration or an applicable exemption from U.S. registration requirements.
About Bluestone Resources Inc.
Bluestone Resources is a mineral exploration and development company that is focused on advancing its 100%-owned Cerro Blanco Gold and Mita Geothermal projects located in Guatemala. A Feasibility Study on Cerro Blanco returned robust economics with a quick pay back. The average annual production is projected to be 146,000 ounces per year over the first three years of production with all-in sustaining costs of $579/oz (as defined per World Gold Council guidelines, less corporate general and administration costs). The Company trades under the symbol “BSR” on the TSX Venture Exchange and “BBSRF” on the OTCQB.
On Behalf of Bluestone Resources Inc.
“Darren Klinck”
Darren Klinck | President, Chief Executive Officer & Director
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
- Published in Bluestone Resources, Mining, News Home
Cannara Biotech – www.shopCBD.com
Cannara Biotech (CSE:LOVE)(FRA:8CB)
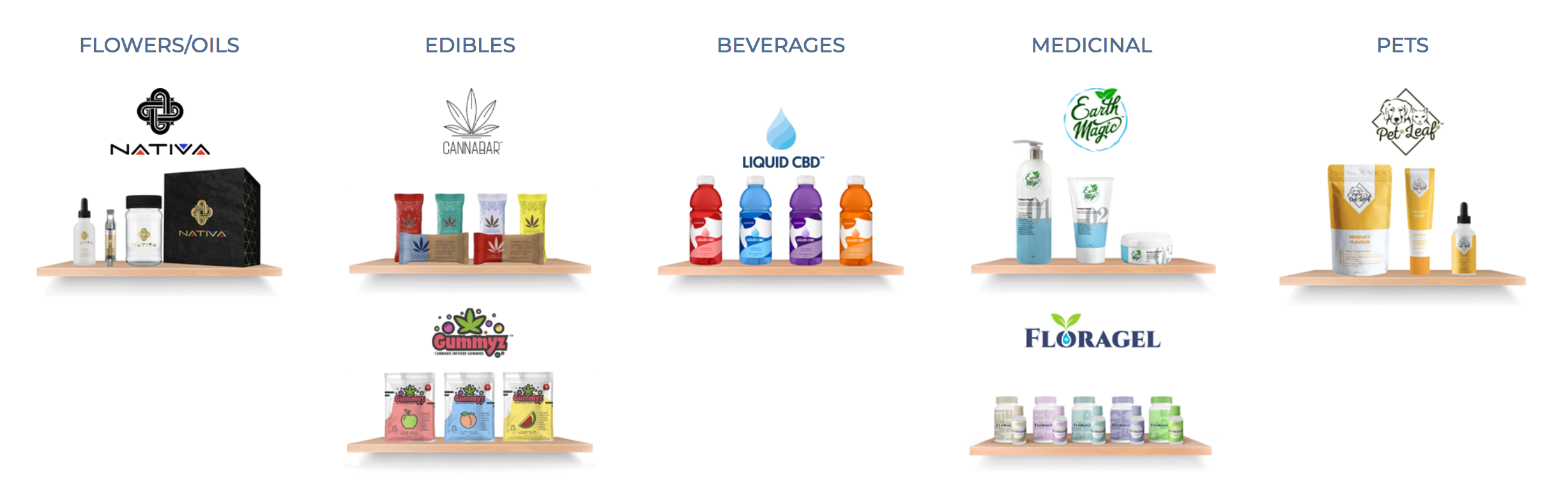
Headquartered in Montreal, Cannara Biotech is entering the U.S. THC-CBD market, through a subsidiary, with an on-line e-commerce platform called shopCBD.
“We are taking a leadership role by creating a hub for vendors to showcase their products and consumers to purchase hemp-CBD products in a fast and easy way. We want to become the Amazon of CBD,” – ZOHAR KRIVOROT – CEO and Founder
Highlights
- Zohar Krivorot, president and chief executive officer of Cannara Biotech is a 15-year veteran in the tech and on-line industries.
- Cannara Biotech is building the largest indoor cannabis cultivation facility in Quebec and one of the largest in Canada
- shopcbd.com is a platform that will showcase hemp-CBD retail products including tinctures, oils, capsules, body care, vape cartridges and pet-related CBD infused treats
- The indoor facility offers lower risk of airborne infections, mold/mildew and produces a higher grade of cannabis
- The new e-commerce platform will showcase retail products from hemp-CBD manufacturers for the U.S. consumer market
- shopCBD will provide a user-friendly on-line experience where consumers can purchase, review and compare a variety of CBD products
Trending
- With the recent passing of the 2018 Farm Bill in the U.S., CBD represents an emerging sector with many vendors seeking national reach to U.S. customers.
- The company’s strategy is to offer a wide variety of products from leading CBD brands, with competitive pricing and delivery times across the U.S.
- The rise in popularity for CBD products is driven by consumers seeking natural-based health and wellness solutions to treat various ailments from inflammation, anxiety to insomnia amongst many others.
- The hemp-derived CBD market is expected to reach $22-billion (U.S.) by 2022 according to industry analysts at Brightfield Group.
Alongside the e-commerce, on-line platform, Cannara Biotech will have on-site independent THC and CBD brands, following the adoption of regulations on cannabis-infused edibles and beverages, that will produce and ship from the facility. Cannara Biotech’s facility will be a multi-purpose facility that will cultivate recreational cannabis and produce cannabinoid-infused products including edibles, cosmetics, pet products and beverages. These products will be shipped to stores and retail outlets in Canada and to international markets.
Cannara Biotech went public in Q1 of this year and is being traded on the CSE under the ticker “LOVE” and on the FRA under the ticker “8CB”. As of February 12th, 2019, LOVE closed at .22.
—
Isabelle Arsenault
Media Relations
MomentumPR
- Published in Cannara, Marijuana, Medical Marijuana
