Cannara Biotech – Quebec’s largest cultivation facility
Cannara Biotech (CSE:LOVE)(FRA:8CB)

Headquartered in Montreal, Cannara Biotech is building the largest indoor cannabis cultivation facility in Quebec and one of the largest in Canada, a modern and secure 625,000-square-foot facility in Farnham, Quebec.
“Being in Quebec allows us to take advantage of the low cost of electricity and gives us a cost advantage over our competitors.” – ZOHAR KRIVOROT – CEO and Founder
Highlights
- Some 100,000 kg of cannabis production is expected per year once all 3 phases are operational. Today, phase 1 is being built with completion expected end of Q2 2019. Once completed, yield is expected at 15,000 kg.
- Cannara Biotech has raised $55M to date, has acquired 19 provisional patents for product exclusivity, and has developed 8 assorted THC/CBD infused product brands to add to their offerings.
- Low electricity cost in Quebec as well as reduced labour cost compared to the rest of Canada gives the Company a cost advantage over their competitors.
- Independent THC and CBD brands will be produced on-site accelerating speed to market.
- The Company will leverage the facility with joint-ventures to produce edibles, pet-products, cosmetics and beverages once the processing license is secured which is estimated for the end of 2019.
- The advantage of growing in an indoor facility offers lower risk of airborne infections, mold/mildew and produces a higher grade of cannabis.
- Cannara Biotech is building an online e-commerce platform (shopCbd.com) that will serve as a podium and distribution channel for US-CBD brands for the American market.

Closer look
Phase 1 will provide 130,000ft2 of growing space. Growing is expected to commence in the fall of 2019 with an estimated yield of 15,000kg.
Cannara Biotech raised $55 million from private investors during the introduction of this project and still have over $40 million in the bank with Phase 1 fully funded.
Phase 2 forecasted to be running by 2020 is 205,000ft2 and likely to yield 38,000kg per year. Phase 3 aiming to be operational in 2022 is 277,000ft2 and expected to produce 55,000kg per year. Cannara Biotech is looking to produce over 100,000kg per year once fully operational.
Currently, 300 sq. ft. space for Phase 2 is being rented by occupants that contribute to a rental revenue of $2 million per year. These earnings pay the monthly fee for the whole facility.
Cannara Biotech’s strategy is also to maximize on the low electricity rates Quebec has with its Hydro and low labour rates in Quebec.
Growing indoor cannabis often results in a higher grade of cannabis. Growing in a controlled environment, protected from many outdoor ailments and mildew creates a more consistent and desirable product for buyers and consumers.
Alongside selling cannabis to intermediaries who will transform the crop into their desired product, Cannara Biotech will have on-site independent THC and CBD brands, following the adoption of regulations on cannabis-infused edibles and beverages, that will produce and ship from the facility. Cannara Biotech’s facility will be a multi-purpose facility that will cultivate recreational cannabis and produce cannabinoid-infused products including edibles, cosmetics, pet products and beverages. These products will be shipped to stores and retail outlets in Canada and to international markets.
Cannara Biotech went public in Q1 of this year and is being traded on the CSE under the ticker “LOVE” and on the FRA under the ticker “8CB”. As of February 12th, 2019, LOVE closed at .22.
—
Isabelle Arsenault
Media Relations
MomentumPR
Canada Cobalt Ramps Up Drilling to Build on New Castle East Discovery Next to Three Past Producers
Momentum Public Relations
Press Release: February 19, 2019
Canada Cobalt Works Inc. (TSXV: CCW) (OTC: CCWOF) (Frankfurt: 4T9B) (the “Company” or “Canada Cobalt”) is pleased to announce that nearly 2,000 meters of drilling in holes CS-18-15 through CS-18-18 has intersected a pyrite-rich Archean gold system (Castle East) in association with newly-discovered fault structures and a strong geophysical signature approximately 1.5 km east of the Castle mine. Drilling of hole CS-18-19 begins during the week of February 18.
Notably, intense quartz veining, abundant fine-grained disseminated pyrite and multiple styles of alteration in CS-18-18 suggests geologists are tracking a robust mineralized system as they target a potentially significant new grassroots discovery that is strengthening toward the west. A 725-meter long, east-west trending IP anomaly with an associated chargeability halo was detected in this area.
Initial assay results for the first two drill holes, including the CS-18-16 wedge hole that was completed to a depth of 586 meters, are expected shortly and will be released as soon as they are received and reviewed.
The Gowganda Silver Camp, which featured three main producers including Castle, has never been systematically explored for its gold, base metal and PGE potential outside of the Nipissing diabase which hosts the high-grade silver-cobalt mineralization in the area.
The Castle mine is approximately 75 kilometers southeast of the Kirkland Lake Gold Camp and just 15 km east of iMetal Resources’ Gowganda West Project where first-ever drilling contiguous to Pan American Silver’s gold deposit is in progress.
Re-2OX And Underground Program (Phases 1 and 2)
Canada Cobalt eagerly anticipates updating shareholders with respect to Re-2OX developments as well as more Phase 1 results and Phase 2 progress for the underground program at the Castle mine. Phase 2 is being strategically expanded through new initiatives that have included the involvement of SGS Lakefield and Wood, building on the success of Phase 1 as the Company leverages its unique advantages in Canada’s cobalt heartland.
Qualified Person
The technical information in this news release was prepared under the supervision of Frank J. Basa, P.Eng., Canada Cobalt’s President and Chief Executive Officer, who is a member of Professional Engineers Ontario and a qualified person in accordance with National Instrument 43-101.
About Canada Cobalt Works Inc.
Canada Cobalt is a pure play cobalt company focused on its past producing Castle mine in the Northern Ontario Cobalt Camp, Canada’s most prolific cobalt district. With underground access at Castle, a recently installed pilot plant to produce cobalt-rich gravity concentrates on site, and a proprietary hydrometallurgical process known as Re-2OX for the creation of technical grade cobalt sulphate as well as nickel-manganese-cobalt (NMC) formulations, Canada Cobalt is strategically positioned to become a vertically integrated North American leader in cobalt extraction and recovery.
“Frank J. Basa”
Frank J. Basa, P. Eng.
President and Chief Executive Officer
- Published in Canada Cobalt Works, Mining, News Home
Bluestone Files Technical Report for its Cerro Blanco Gold Project
Momentum Public Relations
Press Release: February 14, 2019
Bluestone Resources Inc. (TSXV: BSR) (OTCQB: BBSRF) (“Bluestone” or the “Company”) is pleased to announce that the 43-101 feasibility study technical report for the Cerro Blanco Gold Project in Guatemala has been filed and is available for review under the Company’s profile on SEDAR at www.sedar.com and on the Company’s website at www.bluestoneresources.ca.
The recently released feasibility study results (see News Release dated January 29, 2019) demonstrate the project’s robust, rapid pay-back, high-grade, development-ready underground gold mine with a modest capital expenditure and superior economics.
About Bluestone Resources
Bluestone Resources is a mineral exploration and development company that is focused on advancing its 100%-owned Cerro Blanco Gold and Mita Geothermal projects located in Guatemala. A Feasibility Study on Cerro Blanco returned robust economics with a quick pay back. The average annual production is projected to be 146,000 ounces per year over the first three years of production with all-in sustaining costs of $579/oz (as defined per World Gold Council guidelines, less corporate general and administration costs). The Company trades under the symbol “BSR” on the TSX Venture Exchange and “BBSRF” on the OTCQB.
On Behalf of Bluestone Resources Inc.
“Darren Klinck“
Darren Klinck | President, Chief Executive Officer & Director
For further information, please contact:
Bluestone Resources Inc.
Stephen Williams | VP Corporate Development & Investor Relations
Phone: +1 604 646 4534
iinfo@bluestoneresources.ca
www.bluestoneresources.ca
- Published in Bluestone Resources, Mining, News Home
Crop Receives 2019 Production and Extraction Licenses for Its 2,115 Acre Nevada Hemp Operations
Momentum Public Relations
Press Release: February 14, 2019
CROP INFRASTRUCTURE CORP. (CSE: CROP) (OTC: CRXPF) (Frankfurt: 2FR) announced today that its 49% owned subsidiary, Elite Ventures (“Elite”) reported that Hempire, the company’s Nevada tenant, has been issued with a 2019 hemp handler, hemp nursery and hemp grower licenses covering 1,350 irrigated acres on its 2,115 acres of owned or leased CBD focused lands.
A hemp handler is defined as a person who is registered by the licensing department to receive industrial hemp for processing into commodities, products or agricultural hemp seed.
The company chose to apply for its hemp grower licence, nursery and hemp handler licence in anticipation of its lab extracting and producing CBD isolate with a throughput capacity of one ton per day of raw biomass. Furthermore, the company increased the acreage size covered by its hemp licences from 240 acres of cultivation in 2018 to 1,350 acres of irrigated cultivation for 2019.
50,000 square feet of greenhouses are being erected to complement the plant starts coming out of the recently completed genetics laboratory.
CROP CEO, Michael Yorke, stated: “With the increased size and operating range of our tenant’s licenses, CROP’s Nevada CBD operations are emerging into a truly world class operation. Our land holdings, micropropagation team, strategies, drying and extraction capabilities and supply agreements will enable us to operate from seeds to isolate to sales in 2019.”
About CROP
CROP is publicly listed on the CSE and trades under the symbol “CROP”. CROP is focused on cannabis branding and real estate assets. CROP’s portfolio of projects includes cultivation properties in California, two in Washington State, a 1,000-acre Nevada Cannabis farm, 1,865 acres of CBD farms, extraction in Nevada and joint ventures on West Hollywood and San Bernardino dispensary apps with international focuses in Jamaica and Italy.
CROP has developed a portfolio of assets including Canna Drink, a cannabis infused functional beverage line, US and Italian distribution rights to over 55 cannabis topical products and a portfolio of 16 Cannabis brands. CROP’s infrastructure has over 150,000 sq. ft of built canopy and over 2,900 acres of real estate.
Disclaimer for Forward-Looking Information
Certain statements in this press release are forward-looking statements are not based on historical facts, but rather on current expectations and projections about future events and are therefore subject to risks and uncertainties which could cause actual results to differ materially from the future results expressed or implied by the forward-looking statements. In addition, marijuana remains a Schedule I drug under the United States Controlled Substances Act of 1970. Although Congress has prohibited the US Justice Department from spending federal funds to interfere with the implementation of state medical marijuana laws, this prohibition must be renewed each year to remain in effect. These statements generally can be identified by the use of forward-looking words such as “may”, “should”, “could”, “intend”, “estimate”, “plan”, “anticipate”, “expect”, “believe” or “continue”, or the negative thereof or similar variations. Forward-looking statements in this news release include statements regarding the expected returns from the Nevada Project; the technological effects of Nevada Project; the intention to expand its portfolio; and execute on its business plan. Such statements are qualified in their entirety by the inherent risks and uncertainties surrounding the regulatory and legal framework regarding the cannabis industry in general among all levels of government and zoning; risks associated with applicable securities laws and stock exchange rules relating to the cannabis industry; risks associated with maintaining its interests in its various assets; the ability of the Company to finance operations and execute its business plan and other factors beyond the control of the Company. Such forward-looking statements should therefore be construed in light of such factors, and the Company is not under any obligation, and expressly disclaims any intention or obligation, to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by applicable law.
The CSE has not reviewed, approved or disapproved the content of this press release.
Company Contact
Michael Yorke – CEO and Director
E-mail: info@cropcorp.com
Website: www.cropcorp.com
Phone: (604) 484-4206
View original content:http://www.prnewswire.com/news-releases/crop-receives-2019-production-and-extraction-licenses-for-its-2-115-acre-nevada-hemp-operations-300795430.html
- Published in Business, CROP Infrastructure, hemp, Marijuana, News Home
“Heat Engine” May Link Growing Pipeline of Targets At Crystal Lake’s Newmont Lake Project
Momentum Public Relations
Press Release: February 12, 2019
Crystal Lake Mining Corporation (TSXV: CLM, the “Company” or “CrystalLake“) is pleased to provide an update on the latest understanding of the mineral system model for the variety of occurrences and exploration targets within the 430 sq. km Newmont Lake Project along the western flank of the Eskay Rift – one of the largest land packages in this prolific district.
- Ongoing reinterpretation by Crystal Lake’s technical team of historical and recent released data indicate the potential for a linkage between the historic Northwest gold zone, the Ken-Glacier-O’Neill mineral zone (KGO) approximately 3 km to the north, and other adjacent occurrences along this trend. These exploration targets show potential to host high-grade systems and may be genetically linked to a cluster of granitoid intrusions that comprise part of a “Heat Engine” along the Newmont Lake Graben structure in the heart of the property;
- Within this geological framework, Crystal Lake has a focus on the discovery of mineral deposit types including but not limited to high-grade gold, copper-gold porphyry, skarn and volcanogenic massive sulphide types;
- A total of 22 separate mineral occurrences have been outlined historically across the project area. Rapidly receding snowfields, advances in technology, and a much greater understanding of the regional geology leads the Crystal Lake team to believe that the Newmont Lake Project will emerge as a “jewel” of the Golden Triangle in 2019;
- Crystal Lake looks forward to providing a series of updates including more results from last fall’s programs, notably from the Company’s first drilling at Burgundy Ridge and the Northwest zone, as the month progresses.
Dr. Peter Lightfoot, Technical Advisor for Crystal Lake who was part of the team responsible for the discovery of the Golden Triangle’s first nickel-copper-rich massive sulphide system less than 15 km from the southern border of the Newmont Lake Project, commented: “There are vast tracts of unexplored and under-explored ground within the Newmont Lake Project to feed an already large pipeline of quality targets rarely seen on one property. Our mission is to focus on the targets we interpret to have the highest immediate potential. This has me and our discovery team extremely excited for 2019.”
Crystal Lake has an option to earn a 100% interest in the Newmont Lake Project from Romios Gold Resources (TSXV: RG) – refer to September 24, 2018, and December 3, 2018, news releases.
Qualified Person
The technical information in this news release has been reviewed and approved by Dr. Peter C. Lightfoot, P.Geo., a Qualified Person responsible for the scientific and technical information in this news release under National Instrument 43-101 standards.
About Crystal Lake Mining
Crystal Lake Mining is a Canadian-based junior exploration company focused on building shareholder value through high-grade discovery opportunities in British Columbia and Ontario. The Company has an option to earn a 100% interest in the Newmont Lake Project, one of the largest land packages among juniors in the broader Eskay region in the heart of Northwest B.C.’s Golden Triangle.
On behalf of The Board of Directors of Crystal Lake Mining Corporation
Richard Savage, President & CEO
This news release may contain certain “forward looking statements”. Forward-looking statements involve known and unknown risks, uncertainties, assumptions and other factors that may cause the actual results, performance or achievements of the Company to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. Any forward-looking statement speaks only as of the date of this news release and, except as may be required by applicable securities laws, the Company disclaims any intent or obligation to update any forward-looking statement, whether as a result of new information, future events or results or otherwise.
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
SOURCE Crystal Lake Mining Corporation
View original content to download multimedia: http://www.newswire.ca/en/releases/archive/February2019/12/c1942.html
- Published in Business, Crystal Lake Mining, Mining, News Home
Significant gold values extending over more than one km for Stelmine
Momentum Public Relations
Press Release: February 12, 2019
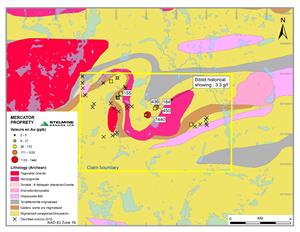
Stelmine Canada (« Stelmine ») (STH-TSXV) submits the results of its exploration campaign conducted on the Mercator property located less than 85 km NW of the Courcy flagship property. Gold values provided by three grab samples (Figure 1) yielded:
|
|||||
- 7.44 g/t Au with 1090 ppm As
- 0.45 g/t Au with 87 ppm As
- 0.44 g/t Au with 19 ppm As
The mineralized sites form the SW extension of the rusted gold-mineralized Biblain showing identified 1 km away by the MRNQ (3.0 g/t Au; Leclair et al., 1998).
Stelmine geologists visited more than 38 sites and collected 22 grab samples during a 5 day heliborne campaign completed during September 2018. The mineralized zones constitute sulfurized and oxidized metasedimentary sequences (2-10 %; mainly pyrrhotite±pyrite) within amphibolite-rich (grunerite) and garnet-rich layers possibly forming iron formations.
Isabelle Proulx, President and CEO of Stelmine declares: “The Mercator property, although sparsely explored, revealed significant gold values extending for 1 km along strike and associated with iron formations. This motivates Stelmine to pursue further its exploration within this prospective zone.”
Regional geology
The Mercator property is located within the extension of the Opinaca metasedimentary sub-province just south of the western segment of the Ashuanipi sub-province and east of the contact with the La Grande sub-province. Most metasedimentary rocks are constituted of greywackes, arenites, siltstones and iron formations (mostly chert). The rocks are highly metamorphosed and migmatized and are largely represented by paragneiss some of which may be dioritic gneiss. The rocks probably belong to the Hublet Group (Mercator Fm). The main plutonic rocks are constituted of migmatized tonalites revealing 15-35 felsic mobilizates of granitic composition (Figure 1).
Additional information
Stelmine also discloses the issuance, as of February 12, 2019, of a total of 850 000 stock options granted according to the Company’s Option Plan. 700,000 options were granted to directors, executive officers and employee for a period of 5 years. The Company also allocated 150,000 options to a consultant for a period of 1 year. All options are issued at the grant date at an exercise price of 0.15$ and each option may be exercised during each period specified above from the grant date. Subject to approval of the TSX Venture Exchange.
Stelmine is proud to announce the nomination of Julie Lemieux as the Company’s CFO. Ms. Lemieux has been working in the accounting field for more than 22 years. She has been a controller at the company since last year. Previously, she acted for various private companies as a controller and worked for over 5 years at a public company where she gained the experience that adds value to Stelmine. Furthermore, Stelmine announces the resignation of Mr. Paul-André Lemieux, CPA, CA. The Company expresses his gratitude for his implication and meticulous work.
QA/QC Protocol
Stelmine implemented a strict QA/QC protocol in processing all rock samples collected from the Mercator property. The protocol included the insertion and monitoring of appropriate reference materials, in this case high concentration (OREAS-210) and low concentration (OREAS-252) certified gold standards, blanks and duplicates, to validate the accuracy and precision of the assay results. All collected rock samples were put in sturdy plastic bags, tagged and sealed in the field under the supervision of professional geologists. The sample bags were then put in rice pouches and kept securely in a field tent before being sent by floatplane to the city of Wabush. Transport to Actlabs laboratories in Ancaster, Ontario was made by truck from reliable transport companies. Gold was analyzed by fire assay method (Au-FA_ICP) and other trace metals via TD-MS or TD-ICP methods (ME-MS61).
The technical contents of this release were approved by Michel Boily, PhD, geo; a Qualified Person (QP) as defined by National Instrument 43-101.
Follow us on www.Stelmine.com and on our Facebook page (Stelmine Canada) where we uploaded pictures of the Courcy camp and of our crew at work.
About Stelmine
Stelmine is a junior mining exploration company which concentrates its activities in the Province of Québec. Stelmine holds 955 claims spanning 497 km2 on the eastern part of the Opinaca metasedimentary basin, which contains zones with a high potential for gold deposit discovery in geological contexts similar to the one leading to discovery of the Éléonore Mine. Its capital stock consists of 29,648,645 issued and outstanding shares for a current market capitalization of $3.5 million.
Forward-looking statements
The statements set forth in this press release, which describes Stelmine’s objectives, projections, estimates, expectations or forecasts, may constitute forward-looking statements within the meaning of security legislation. Positive or negative verbs such as “believe”, “can”, “shall”, “intend”, “expect”, “estimate”, “assume” and other related expressions are used to identify such statements. Stelmine wishes to state that, by their very nature, forward-looking statements involve risks and uncertainties such that its results, or the measures it adopts, could differ materially from those indicated or underlying these statements or could have an impact on the degree of realization of a particular projection. Major factors that may lead to a material difference between Stelmine’s actual results and the projections or expectations set forth in the forward-looking statement include the risk that the Company does not complete the Placement as anticipated, and such other risks as described in detail from time to time in the reports filed by Stelmine with security authorities in Canada. Unless otherwise required by applicable securities laws, Stelmine disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of the new information, future events or otherwise. The forward-looking information in this release is based on information available as of the date of this release.
The TSX Venture Exchange has not reviewed and does not accept responsibility for the adequacy or accuracy of this release.
For more information :
| Isabelle Proulx President and CEO 418-626-6333 |
Christian Guilbaud Business Development 514-813-7862 |
info@stelmine.com Stelmine.com |
A photo accompanying this announcement is available at http://www.globenewswire.com/NewsRoom/AttachmentNg/1e67a598-7797-4394-8e85-d3a9a96fc866
- Published in Mining, News Home, Stelmine Canada
Tetra Bio-Pharma Provides Update on its Neuropathic and Chronic Pain Drug Development Program
Momentum Public Relations
Press Release: February 12, 2019
Tetra Bio-Pharma Inc. (“Tetra” or the “Corporation“) (TSX VENTURE: TBP) (OTCQB: TBPMF), a global leader in cannabinoid-derived drug development and discovery, today announced accelerated development of its second-generation drug inhalation program for fibromyalgia and uncontrolled pain over the last months.
A Phase 1 study in healthy volunteers using vaporized PPP001 was successfully completed (click here) and the Corporation filed a patent application after unexpectedly discovering the unique composition of cannabis smoke. In July 2018, the Corporation entered into a co-development agreement with Storz & Bickel, widely recognized as the global leader in vaporizer design and manufacturing. Tetra incorporated the vaporizer into its smoke-vapor research program as it aimed to understand how inhaled cannabis relieves pain for patients.
About Neuropathic Pain (Fibromyalgia) and Chronic Pain
Fibromyalgia affects about 4 million U.S. adults or about 2% of the adult populationi. The main symptom associated with the condition is the wide range of pain symptoms that can be deep, sharp, dull, throbbing, or aching, affecting the muscles, tendons, and ligaments around the joints throughout the entire body. Treatments aimed at pain relief are the primary treatment intervention including an increasing use of opioids. Chronic pain is also associated with numerous other medical conditions including rheumatoid arthritis, osteoarthritis, gastro intestinal problems, cancer, surgery, HIV/AIDS. The global fibromyalgia treatment market was valued at approximately $10 billion dollars in 2017ii.
Tetra gained knowledge and understanding as to how smoked and vaporized cannabis works to relieve pain, based on its double-blind randomized placebo-controlled studies of PPP001 administered with the titanium pipe and Mighty Medic vaporizer and the use of smoke-vapor trapping studies to quantify exposure to the respiratory tract. A significant finding of the study was learning how much of each cannabinoid and terpene is delivered to the human body as the result of smoking or vaporizing a dried cannabis product. The key to the success of a smoked cannabis product is closely linked to its speed of delivery to the brain. In addition, the inhalation mode of delivery results in significantly higher maximal plasma levels at the peak of treatment. What does this mean? Essentially, results from our study in humans showed that the peak therapeutic levels show only a 1-minute difference in delivery between the pipe and vaporizer. Combined with the speed, the levels of cannabinoids delivered at this peak of exposure are what results in the benefits seen in patients who suffer from pain.
The pipe or vaporizer delivered important differences in some cannabinoids and terpenes. Tetra is using this insight to accelerate the development of alternative formulations for the treatment of fibromyalgia and uncontrolled chronic pain. This knowledge is incorporated in the filed patent application and based on this, Tetra will use isolates or synthetic-based supplies of cannabinoids and terpenes to develop these prescription cannabinoid-derived medicines.
During the last quarter of 2018, the Corporation began preparations for a research site in the United States to conduct its fibromyalgia clinical trial and we are intent on expanding clinical sites in Canada and in the USA in 2019.
“We previously communicated that after reviewing the human clinical data, Tetra anticipated proceeding into phase 2 clinical trials in fibromyalgia patients,” said Dr. Guy Chamberland, CEO and CSO of Tetra Bio-Pharma. “We are committed to this target and plan to accelerate this development in the USA as well. Tetra has and continues to gain tremendous credibility within the medical community. We earned this credibility because of our clinical studies and exhaustive characterization of the pharmaceutical composition of cannabis-derived smoke and vaporization products. Overall, 2018 was a productive year after completing several clinical trials and we expect 2019 to build on this as we accelerate this program.”
About Tetra Bio-Pharma
Tetra Bio-Pharma (TSX-V: TBP) (OTCQB: TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and development with a Health Canada approved, and FDA reviewed, clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. The Company has several subsidiaries engaged in the development of an advanced and growing pipeline of Bio Pharmaceuticals, Natural Health and Veterinary Products containing cannabis and other medicinal plant-based elements. With patients at the core of what we do, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies.
For more information visit: www.tetrabiopharma.com
- Published in Business, Medical Marijuana, News Home, Tetra Bio Pharma
Mondias Completes Characterization of CELEXT07 that Confirms Its Biostimulant and Antifungal Capabilities and Expands Its Global Market Potential
Momentum Public Relations
Press Release: February 12, 2019
Mondias Naturals Inc (“Mondias” or “NHP“), (TSXV: NHP), announces that the research collaboration with McGill University has led to the identification of CELEXT07 signatory metabolite compounds, antibiotic properties and functional roles in plant physiology. This will lead to the development of several commercial products to meet emerging needs in bio-agricultural and organic markets and offer new chemical free solutions for the cultivation and food storage industries.
The chemical classes of compounds identified in the CELEXT07 extract confirms the botanical agent’s properties associated with plant defense against biotic stress by boosting the plant’s resistance as well as a biostimulant effect on growth. The identification of these compounds supports the previous research demonstrating CELEXT07’s biostimulant and antifungal protective and treatment properties.
Mr. Jean-Philippe Gravel, CEO of Mondias Naturals stated, “These results validate Mondias’ strategy to develop natural products for agricultural markets including post-harvest sub-markets. The CELEXT07 botanical extract will make a significant contribution to the bio-agricultural and organic markets and allow growers to offer horticultural products grown with no chemical agents. We are excited by this advancement since CELEXT07 is an eco-friendly alternative to synthetic fungicides.”
Postharvest diseases caused by storage fungi are responsible for the estimated loss of about 20 to 25% of harvested fruits and vegetables, and will certainly affect Canada’s new cannabis industry. Some fungi may produce mycotoxins that can cause toxicity to both animals and humans. Mycotoxin contaminations can occur in the field and greenhouse, before and after harvest, and also during post-harvest activities and storage. Mondias has been developing its CELEXT07 products to help prevent fungal growth on plants through its proven ability to increase plants’ defense system.
As previously reported in its December 5, 2018 news release, CELEXT07, when used as a soil drench agent, significantly reduced lesions caused by fungi on two greenhouse growing plants, tomato and lettuce, and significantly reduced lesions on detached leaves. Mondias has been expanding its research activities to target food and crop storage. Mr. Jean-Philippe Gravel, CEO of Mondias Naturals stated, “These results suggest that CELEXT07 botanical extract may become a low-risk solution to the new Canadian and USA cannabis agricultural industries.”
The market potential for CELEXT07 is considerable considering that these infections result in annual economic losses between $10 billion to $100 billion worldwide (L. Hua et al. 2018) and that about one-third of the food produced worldwide for human consumption is lost after harvest (Gastavsson et al., 2011).
About Mondias Natural Products Inc. Mondias specializes in the commercialization and development of evidence based botanical products for the health care, bio agriculture and organic markets. The company is already selling both oral and topical botanical agents to help manage unmet medical needs through Holizen Laboratories, one of its divisions. Mondias is also developing botanical based specialty fertilizers for use on household plants, urban gardens, lawns, golf courses, nurseries or greenhouses in collaboration with McGill’s Faculty of Agricultural and Environmental Sciences.
For more information, visit: mondias.ca
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
- Published in Business, Life Sciences, Mondias Natural, News Home, Technology