Mosaic Minerals Announces Closing of Its Offering
Mosaic Minerals Corporation (CSE: MOC) (“Mosaic” or the “Corporation”) is pleased to announce that it has closed its private placement (the “Offering“), pursuant to which it issued 750,000 flow-through common shares (the “FT Shares”) and 1,050,000 common shares units (the “Units”) of the Corporation at a price of $0.16 per FT Share and $0.12 per Unit for aggregate gross proceeds of $246,000.
Each Unit consist of one common share, and one-half of one common share purchase warrant (each whole, a “Warrant”) with each Warrant entitling the holder to purchase one additional common share in the capital of the Corporation for a period of 12 months from the date of closing of the Private Placement, at a purchase price of $0.25 per common share.
In connection with the Offering, the Corporation has paid a finder’s fee of 7% in cash and issued 52,500 finder’s warrants (“Finder’s Warrants”). Each Finder’s Warrant is exercisable to acquire one additional common share at a price of $0.25 per Warrant for a period of twelve months from issuance.
All securities issued in respect of the Offering will be subject to a hold period of four (4) months and a day ending April 22, 2022. The Offering is subject to final approval of the Canadian Securities Exchange (CSE).
The company would like to thank its shareholders for their confidence in the past year and wish them a happy new year.
About Mosaic Minerals Corporation
Mosaic Minerals Corp. is a Canadian mineral exploration company listed on the Canadian Securities Exchange (CSE: MOC) now focusing on the exploration for future strategic Copper, Nickel and Zinc deposits in priority on the Quebec Province territory which have a long and successful history of base metal production mainly in the Rouyn-Noranda, Matagami, Val-d’Or and Chibougamau mining camps.
On Behalf of the Board
M. Jonathan Hamel
President & CEO
- Published in Mosaic Minerals, News Home
Sonoro Announces Closing of $3,000,000 Private Placement and Commencement of 10,000 Meter Drilling Program
Sonoro Gold Corp. (TSXV: SGO | OTCQB: SMOFF | FRA: 23SP) (“Sonoro” or the “Company”) is pleased to announce the closing of its previously announced non-brokered private placement of 16,666,667 units (the “Units”) at $0.18 per unit, for aggregate gross proceeds of $3,000,000 (the “Offering”). Each Unit consists of one Sonoro Common Share and one Share Purchase Warrant. Each warrant entitles the holder to purchase one additional Sonoro Common Share for a period of two years from the closing date at an exercise price of $0.30 per share.
All securities issued and issuable in connection with the Offering will be subject to a 4-month plus one day hold period ending April 21, 2022. The Offering has received conditional acceptance from the TSX Venture Exchange but remains subject to receipt of final approval from the TSX Venture Exchange.
In connection with the Offering, the Company entered into finder’s fee agreements with iA Capital Markets, PI Financial Corp., Haywood Securities Inc., IBK Capital Corp. and GloRes Securities Inc. (collectively, the “Finders” and each, a “Finder”) pursuant to which the Company paid to each Finder:
- at the election of the Finder, either a cash finder’s fee or Units equal to a maximum 7% of the gross proceeds raised from subscribers introduced to the Company by the Finder, and
- non-transferable finder’s warrants (the “Finder’s Warrants”) equal in number to 7% of the gross proceeds raised from subscribers introduced to the Company by the Finder. Each Finder’s Warrant entitles the Finder to purchase one common share in the capital of the Company at a price of $0.30 for a period of two years following the closing of the Offering.
In total, the Company paid $47,420.30 in Finder’s fees and 263,447 in non-transferable Finder’s Warrants.
Directors and Officers of the Company participated in the Offering by subscribing for 3,011,461 Units, constituting a related party transaction pursuant to TSX Venture Exchange Policy 5.9 and Multilateral Instrument 61-101 – Protection of Minority Security Holders in Special Transactions (“MI 61-101”). The Company relied on Section 5.5(a) of MI 61-101 for an exemption from the formal valuation requirement and Section 5.7(1)(a) of MI 61-101 for an exemption from the minority shareholder approval requirement of MI 61-101 as the fair market value of the transaction did not exceed 25% of the Company’s market capitalization.
The net proceeds from the Offering will be used primarily to fund a 10,000-meter drilling campaign now underway at the Company’s Cerro Caliche Gold Project in Sonora, Mexico and will also be used for concession maintenance and general working capital.
As announced on October 25, 2021, the drilling program will focus on expanding the project’s oxide gold mineralization included in the updated Resource Estimate announced September 15, 2021, as well as the potential higher-grade targets recently identified by a surface sampling program. Previously announced sample assay results returned grades between 2.5 and 14.3 grams of gold per tonne and revealed multiple oxide gold mineralized “windows” with strong stockwork quartz veining and broad zones of gold and silver mineralization. Drilling will also focus on extending mineralization in the southeast region of the concession that was excluded from the updated resource estimate due to lack of required drilling density.
Sonoro’s President and CEO, Kenneth MacLeod, stated, “We are excited to have commenced the next phase of drilling and anticipate another successful program. The wealth of technical data amassed to date suggests the potential to increase the resource within the project’s known mineralized structures as well as the potential for higher grade and extended mineralized structures identified through scout drilling and surface sampling.”
Sonoro’s Chairman of the Board, John Darch, added, “I believe participation by Sonoro’s directors and officers for over 18% of the current financing is a clear demonstration of our confidence in the success of the Cerro Caliche Project. I am also very pleased we have resumed drilling and are assessing the potential for higher-grade near-surface gold and silver mineralization. Increasing the size and grade of the resource could potentially extend the life of the proposed Heap Leach Mining Operation (HLMO) thereby materially improving the economics for the conceptual 15,000 tonnes per day HLMO.”
About Sonoro Gold Corp.
Sonoro Gold Corp. is a publicly listed exploration and development Company holding the near-development-stage Cerro Caliche project and the exploration-stage San Marcial project in Sonora State, Mexico. The Company has highly experienced operational and management teams with proven track records for the discovery and development of natural resource deposits.
| On behalf of the Board of SONORO GOLD CORP. | |
| Per: | “Kenneth MacLeod” |
| Kenneth MacLeod | |
| President & CEO |
For further information, please contact:
Sonoro Gold Corp. – Tel: (604) 632-1764
Email: info@sonorogold.com
- Published in Mining, News Home, Sonoro Gold
Usha Resources Closes Non-Brokered Private Placement and Raises $1,889,130.80 in Quarter
Usha Resources Ltd. (“USHA” or the “Company”) (TSXV:USHA)(OTCQB:USHAF) is pleased to announce that, subject to final approval of the TSX Venture Exchange (the “TSXV“), it has closed the final tranche of its non-brokered private placement (the “Private Placement“). In total, 3,414,335 units (the “Units“) were issued at $0.30 per Unit in both tranches raising gross proceeds of $1,024,300.40.
Each Unit consists of one common share (a “Share“) in the capital of the Company and one-half of one transferable Share purchase warrant (each whole warrant a “Warrant“) with each whole Warrant exercisable at $0.45 per Share for a period of two (2) years from the date of closing of the Private Placement, subject to an accelerated expiry if the closing trading price of the Company’s Shares is greater than $0.75 per Share for a period of 10 consecutive trading days (the “Acceleration Event“), the Company will give notice to the holders of the Acceleration Event and the warrants will expire 30 days thereafter.
All securities issued in the Private Placement are subject to a four-month and one-day hold period and the TSXV hold period. The Company paid finders’ fees totaling $43,421.01 cash and 144,737 non-transferable finder warrants (the “Finder Warrants“) to PI Financial Corp., Haywood Securities Inc., and Raymond James Ltd. in accordance with applicable securities laws in total in both tranches of the Private Placement. The Finder’s Warrants are exercisable on the same terms as the Warrants issued in the Private Placement.
The Company has now raised $1,889,130.80 in the current quarter inclusive of the gross proceeds from Warrants exercised during the quarter, greatly strengthening its balance sheet moving forward as it continues to develop its Lost Basin Gold-Copper Project located in Mohave County, Arizona through ongoing exploration.
The Company also wishes to advise that further to its previous news release, it continues to work with the vendors to close its acquisition of 1236598 B.C. Ltd. (“1236“) through a share exchange agreement (the “Share Exchange Agreement“). 1236has an option to acquire a 100% interest in a Copper-Cobalt Property (the “Property“) located in Silver Bow and Madison Counties, Montana, subject to a 2% net smelter returns royalty, where over 10,000 metres of historical drilling, including by majors such as BHP, Cominco, Homestake, Phelps Dodge and Rio Tinto, have identified the following drill and trench results[1]:
- 1.2% copper, 0.036% cobalt, and 200 ppb gold over 11.7 metres of massive sulfides
- 0.15% to 0.3% cobalt and up to 1.14% copper over 96.3 metres (note, only 1 ft was assayed for every 10 ft)
- 1.8% copper and 450 ppb gold over 1.25 metres
- 19.0% zinc over 0.7 metres
- 19.8% zinc over 0.4 metres
- Up to 4.7% copper, 0.07% cobalt, and 2.3 g/t gold in trenches
“We are very pleased with the outcome of our fundraising efforts in the quarter,” stated Deepak Varshney, P.Geo., CEO of the Company. “The proceeds will provide the Company with excellent financial footing and flexibility as we move into 2022. We are grateful for the ongoing support and very much look forward to an exciting year ahead.”
Qualified Person
The technical content of this news release has been reviewed and approved by Mr. Dean Besserer, P.Geol., a qualified person as defined by National Instrument 43-101 Standards of Disclosure for Mineral Projects.
About Usha Resources Ltd.
Usha Resources Ltd. is a Canadian mineral acquisition and exploration company based in Vancouver, BC, Canada. Usha is exploring for commercially exploitable mineral deposits and is currently focused on deposits located in Northwest Ontario, Canada and the Lost Basin Gold Mining District in Mohave County, Arizona, U.S.A. Usha increases shareholder value through the acquisition and exploration of quality precious and base metal properties and the application of advanced state-of-the-art exploration methods. Usha’s portfolio of strategic properties provides diversification and mitigates investment risk.
We seek Safe Harbor.
USHA RESOURCES LTD.
“Deepak Varshney” CEO and Director
For more information, please phone James Berard, Investor Relations, at 778-228-2314, email jberard@usharesources.com, or visit www.usharesources.com.
- Published in Mining, News Home, Usha Resources
Glen Eagle Resources Announces Shares for Debt
Glen Eagle Resources Inc. (the “ Corporation ”) (TSXV:GER) (OTC:GERFF) is pleased to announce that it agreed to issue 4,045,066 Shares (the ” Debt Shares “) on a “shares-for-debt” basis in settlement of debts of $250,000 (the ” Debt “) owing to Mr. Eric Sprott and Mr. Denis Lavigueur (the “ Creditors ”). The Debt Shares are being issued at a deemed price of $0.0821 per share, in accordance with the policies of the TSX Venture Exchange (the ” TSX-V “).
The issuance of the Debt Shares to the Creditors will constitute a “related party transaction” within the meaning of the TSX-V Policy 5.9 (the ” Policy “) and Multilateral Instrument 61-101 – Protection of Minority Security Holders in Special Transactions (” MI 61-101 “) adopted in the Policy. The Company intends to rely on the exemptions from the formal valuation and minority shareholder approval requirements of MI 61-101 contained in sections 5.5(a) and 5.7(1)(a) of MI 61-101 as neither the fair market value (as determined under MI 61-101) of the Debt Shares, nor the Debt, exceeds 25% of the Company’s market capitalization (as determined under MI 61-101).
The issuance of the Debt Shares is subject to the approval of the TSX-V. Any Debt Shares issued will be subject to a hold period which will expire on the date that is four months and one day from the date of issue.
ABOUT GLEN EAGLE RESOURCES INC.
Glen Eagle Resources Inc. is a small producer and an exploration company of precious metals in Canada and the central America area.
For more information, please contact:
Jean Labrecque
President
1-514-808-9807
- Published in Glen Eagle, Mining, News Home
Sirona Biochem Antiviral Compounds to be Tested Against SARS-CoV-2 by Top US Testing Facility
Sirona Biochem Corp. (TSXV: SBM) (FSE: ZSB) (OTC: SRBCF) (“Sirona“) announces the advancement of 20 antiviral compounds to In Vitro testing. The compounds will be shipped to the Utah State University’s Institute of Antiviral Research for evaluation against the SARS-CoV-2 virus (COVID-19).

Sirona’s French subsidiary, TFChem, has created a novel and patentable library of 20 compounds that are designed to target the COVID-19 virus by targeting the host cell and not the virus itself. When a virus enters a cell, it induces a “cytopathic effect” which refers to structural changes in the host cell, that allow the virus to replicate. In SARS-CoV-2, the virus causes plaque-like effects in the human cells of the lung. Mechanisms such as cell fusion, apoptosis (death) and destruction of epithelium integrity are all observed in the plaque regions (Nature Communications, 2020, https://www.nature.com/articles/s41467-020-17796-z). Sirona’s antiviral compounds are intended to prevent the cytopathic effect from occurring.
The results will be completed in approximately 6 weeks.
“Since the compounds are designed to target the host cell and not the virus itself, we have a unique approach that is based on our proprietary technology. We have seen the number of variants that the SARS-CoV-2 virus has. It will continue to mutate, creating new strains, so there is a strong need for compounds like these that are not targeted to a specific virus,” said Dr. Howard Verrico, CEO of Sirona Biochem. “It is now time to push this program as quickly as possible, adding to a robust and valuable pipeline that will attract the right kind of partners. For this project, we have already been in preliminary discussion with a company that has expertise in antiviral clinical trials. The results from these preliminary studies will be key to moving forward with potential partners in 2022.”
About Sirona Biochem Corp.
Sirona Biochem is a cosmetic ingredient and drug discovery company with a proprietary platform technology. Sirona specializes in stabilizing carbohydrate molecules with the goal of improving efficacy and safety. New compounds are patented for maximum revenue potential. Sirona’s compounds are licensed to leading companies around the world in return for licensing fees, milestone fees and ongoing royalty payments. Sirona’s laboratory, TFChem, is in France and is the recipient of multiple French national scientific awards and European Union and French government grants. For more information, please visit www.sironabiochem.com.
- Published in News Home, Sirona Biochem
Sirona Biochem Announces Engagement of Pullan Consulting to Close Optimal Deal for TFC-1067
Sirona Biochem Corp. (TSX-V: SBM) (FSE: ZSB) (OTC: SRBCF) (“Sirona“) is very pleased to announce that Linda Pullan of Pullan Consulting will be providing strategic direction with the negotiations and licensing of TFC-1067.

Sirona is in the negotiation phase with a global top 10 pharmaceutical company for the rights to TFC-1067. Simultaneously, the company is completing the legal due diligence regarding contracts and patents, which is a straightforward task. The pharma company is advancing on the project as expected and has been transparent with next steps and timelines.
“We are thrilled to have Pullan Consulting on board to assist us with this task. Linda has successfully closed 65 pharma/biotech deals and has advised on many more. She is a renowned professional in the field,” said Howard Verrico, CEO of Sirona Biochem. “We are dealing with a pharmaceutical company that is very experienced in licensing negotiations. The highest level of expertise is needed to obtain the best outcome. We see this level in Pullan Consulting and look forward to working with their team to close an optimal deal.”
About Sirona Biochem Corp.
Sirona Biochem is a cosmetic ingredient and drug discovery company with a proprietary platform technology. Sirona specializes in stabilizing carbohydrate molecules with the goal of improving efficacy and safety. New compounds are patented for maximum revenue potential.
Sirona’s compounds are licensed to leading companies around the world in return for licensing fees, milestone fees and ongoing royalty payments. Sirona’s laboratory, TFChem, is in France and is the recipient of multiple French national scientific awards and European Union and French government grants. For more information, please visit www.sironabiochem.com.
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Sirona Biochem cautions you that statements included in this press release that are not a description of historical facts may be forward-looking statements. Forward-looking statements are only predictions based upon current expectations and involve known and unknown risks and uncertainties. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of release of the relevant information, unless explicitly stated otherwise. Actual results, performance or achievement could differ materially from those expressed in, or implied by, Sirona Biochem’s forward-looking statements due to the risks and uncertainties inherent in Sirona Biochem’s business including, without limitation, statements about: the progress and timing of its clinical trials; difficulties or delays in development, testing, obtaining regulatory approval, producing and marketing its products; unexpected adverse side effects or inadequate therapeutic efficacy of its products that could delay or prevent product development or commercialization; the scope and validity of patent protection for its products; competition from other pharmaceutical or biotechnology companies; and its ability to obtain additional financing to support its operations. Sirona Biochem does not assume any obligation to update any forward-looking statements except as required by law.
SOURCE Sirona Biochem Corp.


View original content to download multimedia: http://www.newswire.ca/en/releases/archive/December2021/16/c3558.html
Contact:
Investor Enquiries: Jonathan Williams, Managing Director, Momentum PR, Phone: 1.450.332.6939, Email: jwilliams@momentumpr.com
- Published in News Home, Sirona Biochem
Glen Eagle Selling $1 Million Dollars of Dore, Including 2000 Ounces of Silver
Glen Eagle Resources (TSXV:GER) (OTC:GERFF) (“Glen Eagle” the “Company”, or “GER”) is pleased to announce that from September to year end, the Company will have sold approximately CDN $1,000,000 dollars in dore bars and projecting to double its sales in Q1-2022 based on the feed and grade discovered at La Esperanza II. The values intersected are summarized for the first time in the table below and detailed in prior News Releases dated November 24 and December 9, 2021.
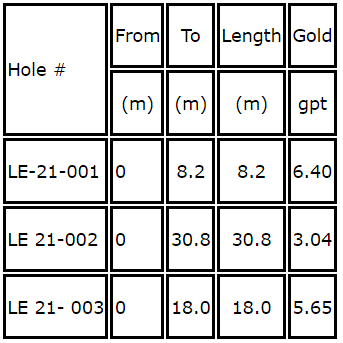
COO Karl Trudeau comments “Having been in Honduras recently to look at Cobra Oro operations, I was pleased by the ergonomics of the plant and its future potential as the inventory of spare parts is slowly being replenished, lowering the risk of plant failures. Some challenges are still lying ahead, but I remain confident that the company will be successful at turning around several aspects of its business, including a very positive ramp up operation still in progress at its gold processing facility in Southern Honduras.”
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.”
- Published in Glen Eagle, Mining, News Home
Sirona Biochem Provides Update on SGLT2 Inhibitor for Type 2 Diabetes in China
Sirona Biochem Corp. (TSXV: SBM) (FSE: ZSB) (OTC: SRBCF) (“Sirona“) announces that it has received notice that the development of Sirona’s SGLT2 inhibitor, TFC-039, will be discontinued by Wanbang Biopharmaceuticals (“Wanbang“) as a treatment for type 2 diabetes in China.

TFC-039 was licensed to Wanbang, a wholly owned subsidiary of Shanghai Fosun Pharmaceutical Group, in 2014. Sirona received a total of USD 1.5 million in up front and milestone payments.
“To this point, Wanbang has remained dedicated to the development of the drug, completing the IND and advancing through and completing its Phase I clinical trials. During 2021, they indicated that they were extensively analyzing the clinical results and that delays were the result of COVID closures and restrictions. This year, Empagliflozin (developed by Boehringer Ingelheim and Eli Lilly) failed to uphold patent protection in China, which created a generic environment for SGLT2 inhibitors. It was reported to us TFC-039 performed well in the clinical trials, however, given the generic situation, it did not make good business sense for Wanbang to continue a lengthy and costly development”, said Dr. Howard Verrico, CEO of Sirona Biochem.
“While this is disappointing, we still see a path forward for TFC-039. We are working on several opportunities (both existing and new) for animal care globally. Our science team is also working on a new therapeutic indication, with potential strong advantages for TFC-039 compared to other compounds of the same class. This new indication has both unmet need and tremendous commercial value. The work done to date by Sirona and Wanbang will provide added value towards these opportunities, and we will also explore future involvement with Wanbang. Further dialogue will be occurring between Wanbang, and Sirona and any material news will be released accordingly.”
About Wanbang Biopharmaceuticals and Fosun Pharmaceuticals
Wanbang Biopharmaceuticals develops, manufactures and sells drugs with indications for chronic disease treatment, antibiotics, and other endocrine diseases in China. Founded in 1981, they are presently headquartered in Xuzhou, China, and are a subsidiary of Shanghai Fosun Pharmaceutical Group. Fosun is a leader in the pharmaceutical industry and regarded as one of the top five domestic pharmaceutical companies in China. For more information on Fosun and Wanbang, please visit www.fosunpharma.com/en.
About Sirona Biochem Corp.
Sirona Biochem is a cosmetic ingredient and drug discovery company with a proprietary platform technology. Sirona specializes in stabilizing carbohydrate molecules with the goal of improving efficacy and safety. New compounds are patented for maximum revenue potential.
Sirona’s compounds are licensed to leading companies around the world in return for licensing fees, milestone fees and ongoing royalty payments. Sirona’s laboratory, TFChem, is located in France and is the recipient of multiple French national scientific awards and European Union and French government grants. For more information, please visit www.sironabiochem.com.
- Published in News Home, Sirona Biochem
- 1
- 2

