Element 79 closes qualifying transaction with Mondias
Momentum Public Relations
Press Release: November 23, 2018
On Nov. 14, 2018, Mondias Natural Products Inc. has closed its previously announced transaction with Element 79 Capital Inc.
The transaction consisted of a transaction that resulted in the reverse takeover of Element by the shareholders of Mondias by virtue of acquiring 100 per cent of the equity interest of Mondias. In connection with the reverse takeover, an aggregate of 41,882,760 common shares were issued at a deemed value of $4,397,689.80. Pursuant to the acquisition of Mondias, an aggregate of 6,882,760 warrants to purchase 6,882,760 common shares at an exercise price of 25 cents per share until June 20, 2020, were granted to Mondias’s existing warrantholders. The transaction constituted Element’s qualifying transaction as such term is defined in Policy 2.4 of the TSX Venture Exchange.
Prior to completion of the transaction, Element consolidated its common shares on a one-for-1.5 basis. As part of the transaction, Element changed its name from Element 79 Capital Inc. to Mondias Natural Products Inc.
As a result of the transaction, Mondias is now a wholly owned subsidiary of Element, and Element will continue the business of Mondias, which consists in the commercialization and development of evidence-based botanical products and natural health products for the health care and bioagriculture markets. For additional information concerning Mondias’s business, please refer to Element’s filing statement with respect to the transaction dated Oct. 29, 2018, which has been filed under Element’s SEDAR profile.
Concurrently to the transaction, Mondias has closed a non-brokered private placement for total gross proceeds of $1,390,725 consisting of the issuance of 13,245,000 postconsolidation common shares of Mondias. No related parties (as such term is defined in applicable laws) were part of the private placement and no subscriber became an insider of Mondias on closing of the private placement.
Prior to the transaction, Element was a capital pool company (as defined under the policies of the TSX-V) and had not commenced commercial operations other than identifying and evaluating potential business acquisitions that would qualify as its qualifying transaction and had no assets other than cash. The TSX-V granted a sponsorship waiver for this qualifying transaction.
Final acceptance of the transaction will occur upon the issuance of a final exchange bulletin by the exchange. Upon issuance of the final exchange bulletin, the company will cease to be a capital pool company and will recommence trading on the exchange as a Tier 2 issuer. Trading in the common shares of the company will begin on the exchange after the final exchange bulletin has been issued under the symbol NHP.
Management of Mondias expects that the final exchange bulletin will be issued within the next week. Pursuant to the exchange’s policies, there will be a delay of two business days between the issuance of the final exchange bulletin and resumption to trading.
Following the completion of the transaction (on a postacquisition basis), the company has a total of 63,135,805 common shares issued and outstanding, as well as: (i) common share purchase warrants exercisable to purchase up to 6,882,760 additional common shares of the company at an exercise price of 25 cents per share until June 20, 2020; and (ii) stock options exercisable to purchase up to 400,402 additional common shares at an exercise price of 22.5 cents per share.
An aggregate of 36,333,334 common shares are subject to escrow pursuant to TSX-V escrow requirements. An aggregate of 35,000,001 common shares issued to principals pursuant to the qualifying transaction will be subject to a Tier 2 value escrow agreement instead of a Tier 2 surplus escrow agreement as previously disclosed in the qualifying transaction filing statement.
As a result of the closing of the transaction, the directors and executive officers of the company are expected to be as shown in the attached table.
Andre Rancourt Chairman of the board, director Frank Palantoni Director Bertrand Venne Director Michel Timperio Director Louis Doyle Director Jean-Philippe Gravel Chief executive officer Sabino Di Paola Chief financial officer Gilles Seguin Corporate secretary
In addition to the company’s directors and officers, Robert Brouillette will also be considered to be an insider of the company by virtue of holding more than 10 per cent of the issued and outstanding securities of the company.
Further details about the transaction and the company as the resulting issuer from the closing of the transaction are available in the filing statement.
- Published in Business, Life Sciences, Mondias Natural, News Home
Tetra Bio-Pharma Seeks Solutions for Second Hand Smoke From Cannabis
Momentum Public Relations
Press Release: November 22, 2018
Tetra Bio-Pharma Inc., a leader in cannabinoid-based drug discovery and development (TSX VENTURE: TBP) (OTCQB: TBPMF), is pleased to update the market on its strategic partnership with Sirius Air Solutions Inc. (previously known as AES) to secure a source of supply for a specialized cannabis smoke air purifying system and a mask designed for law and emergency responders.
The parties are establishing a strategic alliance to develop a medical grade air purifier and mask that will address Health Canada, labour unions and public safety concerns surrounding exposure to the second-hand smoke of cannabis.
Tetra Bio-Pharma expects orders for the mask and air purifier from caregivers, medical institutions, law enforcement agencies and security services around the world.
“This technology will enable the consumers of a smokable cannabis product to take their medication in the privacy of their home or in a medical clinic while being cared for by their families, caregivers or medical personnel,” stated Dr. Guy Chamberland, CEO and CSO of Tetra Bio-Pharma. “We have begun working with law enforcement representatives across the country to design a specialized mask that will alleviate the effects of second-hand smoke from cannabis while performing their duties.”
About Sirius Air Solutions Inc.
Sirius Air Solutions Inc. (SAS) is an ultra-specialized air cleaner and smoke extractor manufacturer aiming to provide efficient answers for the most demanding innovative markets. Moreover, we are specialized in particle ultra filtration of air as well as in the chemical air filtration for contaminants present in gaseous form. Our approach is based on a deep understanding of airborne contaminants in order to design unique devices that consistently strive to exceed the highest standards available to date. Every day, we challenge ourselves to offer the most effective solutions to meet the most demanding situations that are proposed.
About Tetra Bio-Pharma
Tetra Bio-Pharma (TSX-V: TBP) (OTCQB: TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and development with a Health Canada approved, and FDA reviewed, clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. The Company has several subsidiaries engaged in the development of an advanced and growing pipeline of Bio Pharmaceuticals, Natural Health and Veterinary Products containing cannabis and other medicinal plant-based elements. With patients at the core of what we do, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies.
For more information visit: www.tetrabiopharma.com
- Published in Life Sciences, Medical Marijuana, News Home, Tetra Bio Pharma
North Bud Farms Provides Corporate Update
Momentum Public Relations
Press Release: November 22, 2018
North Bud Farms Inc.(CSE: NBUD) (“NORTHBUD” or the “Company”) is pleased to provide shareholders with an update on our corporate activities.
Construction of our Cannabis Production Facility in Low, Quebec
NORTHBUD is pleased to update shareholders that the construction of our cannabis production facility in Low, Quebec remains on schedule as per the timeline provided from our builder NGA Construction. Erecting of the steel structure began on the 19th of November and is expected to take approximately 30 days to complete. We expect the building to be operational in Q1 2019. Please follow our social media channels for video updates of the facility construction (https://vimeo.com/302330795) and branding out reach.
Cannabis Act Application
Migration of the application to the Cannabis Licensing Tracking System (“CLTS”) has begun in collaboration with Cannabis Consulting Inc., one of the leading consultancy firms in the industry.
Update on Janey’s Inc. Acquisition
NORTHBUD and Janey’s continue to work on finalizing the acquisition agreement to include additional product segments. To date Janey’s has fulfilled multiple orders to the Ontario Cannabis Store and intends to expand its offering in the upcoming product calls.
Corporate Initiatives
NORTHBUD wishes to inform shareholders that its Board of Directors has approved management’s request to explore business opportunities in other international jurisdictions.
“This development represents an evolution in our business since going public and we feel it is important to update shareholders and potential shareholders that we will be actively pursuing international opportunities that we believe are complementary to our core business and that should create value for shareholders as we expand NORTHBUD’s global presence,” said Ryan Brown, CEO of North Bud Farms Inc.
About North Bud Farms Inc.
North Bud Farms Inc., through its wholly-owned subsidiary GrowPros MMP Inc. which was acquired in February 2018, is pursuing a license under the Access to Cannabis for Medical Purposes Regulations (ACMPR). North Bud Farms will be constructing a state-of-the-art purpose-built cannabis production facility located on 95 acres of Agricultural Land in Low, Quebec. North Bud Farms will be focused on Pharmaceutical and Food Grade cannabinoid production in preparation for the legalization of edibles and ingestible products scheduled for October 2019.
For more information visit: www.northbud.com.
Neither the Canadian Securities Exchange (the “CSE”) nor its Regulation Services Provider (as that term is defined in the policies of the CSE) accepts responsibility for the adequacy or accuracy of this release.
Forward-looking statements
Certain statements included in this press release constitute forward-looking information or statements (collectively, “forward-looking statements”), including those identified by the expressions “anticipate”, “believe”, “plan”, “estimate”, “expect”, “intend”, “may”, “should” and similar expressions to the extent they relate to the Company or its management. The forward-looking statements are not historical facts but reflect current expectations regarding future results or events. This press release contains forward- looking statements. These forward-looking statements are based on current expectations and various estimates, factors and assumptions and involve known and unknown risks, uncertainties and other factors. Such risks and uncertainties include, among others, the risk factors included in North Bud Farms’ final long form prospectus dated August 21, 2018 which is available under the issuer’s SEDAR profile at www.sedar.com.
FOR ADDITIONAL INFORMATION, PLEASE CONTACT:
North Bud Farms Inc.
Edward Miller
VP, IR & Communications
Office: (855) 628-3420 ext. 3
investors@northbud.com
- Published in Life Sciences, Marijuana, Medical Marijuana, News Home, NorthBud
DIAGNOS Announces Final Closing of Private Placement
Momentum Public Relations
Press Release: November 21, 2018
DIAGNOS Inc. (“DIAGNOS”, the “Corporation” or the “Issuer”) (TSX Venture: ADK) (OTCQB: DGNOF), a leader in early detection of critical health issues through the use of Artificial Intelligence, announces the final closing of a private placement (“Private Placement”) of 20,660,000 units (each a “Unit”) issued at $0.05 per Unit for gross proceeds of $1,033,000 following receipt of required regulatory approvals. The Private Placement was announced on November 6, 2018 and on November 9, 2018.
All monies quoted in this press release shall be stated and paid in lawful money of Canada.
About DIAGNOS
DIAGNOS is a publicly-traded Canadian corporation with a mission of early detection of critical health issues through the use of its Artificial Intelligence (“AI”) tool CARA (Computer Assisted Retina Analysis). CARA is a tele-ophthalmology platform that integrates with existing equipment (hardware and software) and processes at the point of care. CARA’s Artificial Intelligence image enhancement algorithms make standard retinal images sharper, clearer and easier to read. CARA is accessible securely over the internet, and is compatible with all recognized image formats and brands of fundus cameras, and is EMR compatible. CARA is a cost-effective tool for screening large numbers of patients in real-time and has been cleared for commercialization by several regulatory authorities such as Health Canada, the U.S. Food and Drug Administration and the European Union.
Additional information is available at www.diagnos.com and www.sedar.com.
For further information, please contact:
| Mr. André Larente, President | Josh Falle | ||
| DIAGNOS Inc. | Momentum PR | ||
| Tel: 450-678-8882 ext. 224 | Tel: 514-416-4656 | ||
| alarente@diagnos.ca | josh@momentumpr.com | ||
- Published in Diagnos, Life Sciences, News Home, Technology
DIAGNOS Enters into a Shares for Debt Transaction
Momentum Public Relations
Press Release: November 21, 2018
DIAGNOS Inc. (“DIAGNOS” or the “Corporation”) (TSX Venture: ADK) (OTCQB: DGNOF), a leader in early detection of critical health issues through the use of Artificial Intelligence, announces today it has entered into a shares for debt arrangement in which the Corporation is issuing 900,000 common shares (each a “Share”), at a deemed price of $0.05 per Share, in settlement for interests payable in the amount of $45,000 on outstanding unsecured convertible notes (each a “Note”) totaling $900,000.
Satisfying this outstanding indebtedness with shares was undertaken in order to preserve the Corporation’s cash for operational purposes.
Two Note holders eligible to receive common shares as part of the shares for debt transaction are considered “related parties” of the Corporation within the meaning of Multilateral Instrument 61-101 – Protection of Minority Security Holders in Special Transactions (“MI 61-101”). One person related to one Note holder, as well as one other Note holder will each receive 200,000 common shares as part of the transaction. The transaction is exempt from the valuation requirement and the minority approval requirement prescribed in MI 61-101 based on the fact that the fair market value of the related parties’ participations in the shares for debt transaction does not exceed 25% of DIAGNOS’ market capitalization.
Shares issued as part of the shares for debt arrangement are subject to a statutory four-month hold period from the date of issuance ending March 21, 2019.
The issuance of shares is subject to receipt of all required approvals, including the approval of the TSX Venture Exchange, as well as the execution of formal documentation.
All monies quoted in this press release shall be stated and paid in lawful money of Canada.
About DIAGNOS
DIAGNOS is a publicly-traded Canadian corporation with a mission of early detection of critical health issues through the use of its Artificial Intelligence (“AI”) tool CARA (Computer Assisted Retina Analysis). CARA is a tele-ophthalmology platform that integrates with existing equipment (hardware and software) and processes at the point of care. CARA’s Artificial Intelligence image enhancement algorithms make standard retinal images sharper, clearer and easier to read. CARA is accessible securely over the internet, and is compatible with all recognized image formats and brands of fundus cameras, and is EMR compatible. CARA is a cost-effective tool for screening large numbers of patients in real-time and has been cleared for commercialization by several regulatory authorities such as Health Canada, the U.S. Food and Drug Administration and the European Union.
Additional information is available at www.diagnos.com and www.sedar.com.
For further information, please contact:
| Mr. André Larente, President | Josh Falle |
| DIAGNOS Inc. | Momentum PR |
| Tel: 450-678-8882 ext. 224 | Tel: 514-416-4656 |
| alarente@diagnos.ca | josh@momentumpr.com |
- Published in Diagnos, Life Sciences, News Home, Technology
Tetra Bio-Pharma Enters into Binding Proposal with Quantum Pharma Inc.
Momentum Public Relations
Press Release: November 20, 2018
Tetra Bio-Pharma Inc. (“Tetra” or “TBP”), (TSX VENTURE: TBP) (OTCQB: TBPMF), and Quantum Pharma Inc. (“Quantum”) today announced the signing of a binding proposal (the “Proposal“) for exclusive access to a Health Canada licensed facility (i.e., Drug Establishment License; DEL) that enables the company to produce cannabis and cannabinoid drug products for Tetra’s and Panag Pharma’s development activities including the ocular drug formulations. Quantum Pharma owned by Dr. Peter Ford was the GMP manufacturing division of Ford’s Family Pharmacy and Wellness Centre. Their formulation expertise will enable Tetra to develop innovative products for future clinical trials.
The Proposed Transaction is expected to provide Tetra with:
- Exclusive access to a Health Canada licensed facility (DEL) to fabricate cannabis and cannabinoid drug products for Tetra and Panag’s development activities;
- Cannabis and cannabinoid drug product formulation expertise; and
- The ability to manufacture products for all its clinical trials and commercial launches.
Dr. Guy Chamberland, CEO and CSO of Tetra Bio-Pharma stated, “We are very pleased to announce this important development to our shareholders. We have been working with Ford’s Family Pharmacy and Wellness Centre since 2016. Dr. Peter Ford and his team have created several innovative formulations and instruments for the fabrication of Tetra’s prescription drugs. This exclusive agreement not only secures the innovations and production know-how developed for Tetra, but also protects these assets from an acquisition by a competitor. Quantum Pharma will be an exclusive partner to Tetra and Panag enabling us to execute our future planned trials as well as prepare for our product launches.”
Dr. Chamberland further stated, “Tetra has already entered into various agreements to ensure the supply meets the demand for cannabis, THC and CBD-based products with partners including Aphria Inc., True North Cannabis, a USA-based synthetic cannabinoid manufacturer and a phytocannabinoid supplier. With the expected completion of the Panag acquisition, Tetra will become vertically integrated with drug development capabilities that range from discovery all the way through to commercial launch. Our partnerships with Genacol Corporation and Kombucha Baby Brewing Company and its partners provides Tetra with a global sales and distribution platform for the commercialization of OTC drug and wellness products. In addition, Tetra has also established distribution and sales agreements with two pharmaceutical companies for its PPP001 prescription drug product.”
About Tetra Bio-Pharma
Tetra Bio-Pharma (TSX-V: TBP) (OTCQB: TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and development with a Health Canada approved, and FDA reviewed, clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. The Company has several subsidiaries engaged in the development of an advanced and growing pipeline of Bio Pharmaceuticals, Natural Health and Veterinary Health Products containing cannabis and other medicinal plant-based elements. With patients at the core of what we do, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies.
For more information visit: www.tetrabiopharma.com
- Published in Life Sciences, Medical Marijuana, News Home, Tetra Bio Pharma
AREV Brands International Ltd. announces Success in Terpene Blends on 1st Degree Burns and Insect Bites
Momentum Public Relations
Press Release: November 14, 2018
AREV Brands International Ltd. (” AREV ” or the “Company”) ( CSE – AREV ), is pleased to announce AREV Brands International Ltd. has has reached another major milestone in one of its several joint development projects with Alternative Extracts Inc. Further to the research work reported on September 20, 2018, terpenes from non-regulated sources were used to formulate a proprietary composition for burns and insect bites. As noted in peer-reviewed literature, terpene synthesis is vital for cannabinoid synthesis as it provides the GPP molecule for CBGA synthesis.
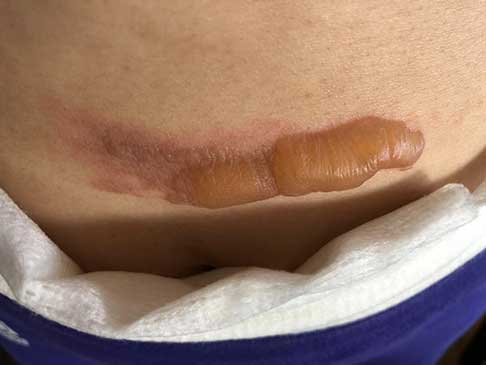
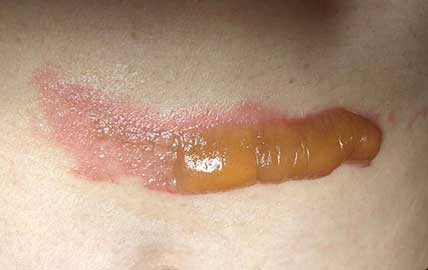
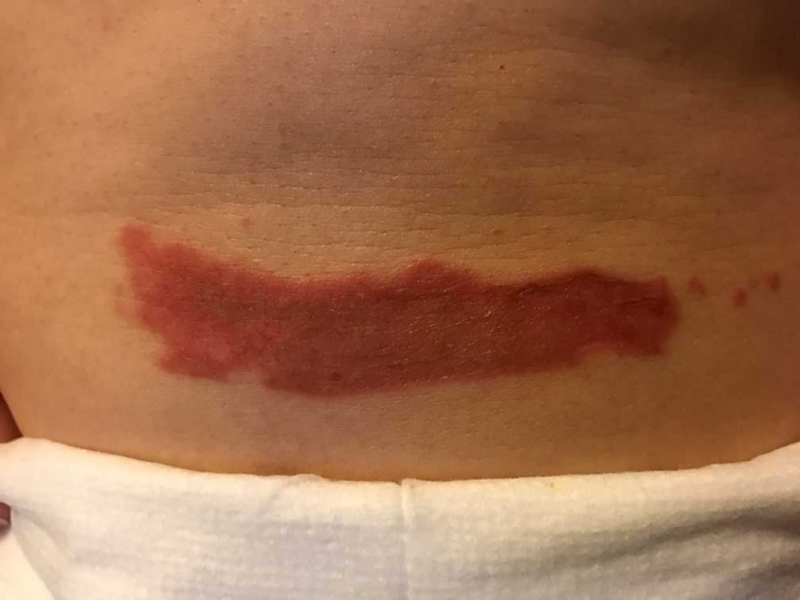
As evidenced in these images (which was first taken within 18 hours of the burn accident, and in 24-hour succession thereafter), the blisters from the burn were completely eliminated within 4 days from the first application. The patient in this case is a 50-year old woman who has a known history of keloid formation. It is noted that the patient did not develop any infection.
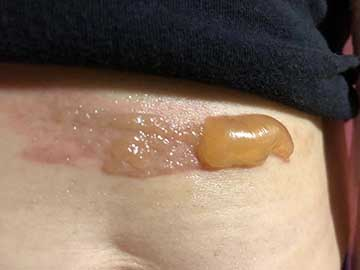
AREV extracted the valuable terpenes from selected non-regulated plant materials and exudates from trees using its extraction systems that it acquired from Alternative Extracts Inc. The composition was formulated in collaboration between AREV and AEI. The mechanism of the composition is believed to be the result of the poly-molecular function of the terpenes.
The company cautions, that although these results are encouraging, a wider sample population is required. A formal study will be conducted in the coming months.
Burn data from World Health Organization shows that:
- – In India, over 1,000,000 people are moderately or severely burnt every year.
– Nearly 173,000 Bangladeshi children are moderately or severely burnt every year.
– In Bangladesh, Colombia, Egypt and Pakistan, 17% of children with burns have a temporary disability and 18% have a permanent disability.
– Burns are the second most common injury in rural Nepal, accounting for 5% of disabilities.
– In 2008, over 410,000 burn injuries occurred in the United States of America, with approximately 40,000 requiring hospitalization.
Mike Withrow commented “Although we are pleased to have been able to minimize the pain and scarring in this burn patient, the results from the use of our proprietary blend of natural compounds warrants further testing and trials.”
Dr. Adrian Wade, Chief Science Officer, of Alternative Extracts Inc. commented, “This collaboration continues to produce highly effective compounds and formulations. Terpenes have demonstrated to play an important role in an increasing number of natural health and medical products.”
Seek safe harbor and forward looking statements.
For further information, contact Mike Withrow, mike.withrow.vn@gmail.com, (778) 896-6536?.
On behalf of the Board,
Mike Withrow
CEO & Director
About AREV Brands International Ltd.
AREV Brands International Ltd. (“AREV”) produces and delivers functional compounds and ingredients from its world-class extraction systems. AREV is revolutionizing the current delivery method of terpenes, cannabinoids and flavonoids. These premium ingredients and formulations are used in products targeted for sale in the natural health, medical, functional food, nutraceutical, sport nutrition and bioceutical markets. AREV innovates through extraction to produce extracts from specific selected plant and exude from trees that address 5 areas of health including Anxiety, Pain Management, Insomnia, Central Nervous System Disorders & Libido.
- Published in AREV Nutrition Sciences, Business, Life Sciences, Medical Marijuana, News Home, Technology
DIAGNOS Announces Today the Addition of a Strategic Partner in the Country of Mexico
Momentum Public Relations
Press Release: November 13, 2018
DIAGNOS Inc. (“DIAGNOS” or “the Corporation”) (TSX Venture:ADK), (OTCQB: DGNOF), a leader in early detection of critical health issues through the use of Artificial Intelligence (AI), announces today the addition of a strategic partner in the country of Mexico. This agreement will enlarge the footprint coverage of DIAGNOS’ offering for monitoring diabetic patients through our first partner in that country.
“Today we all (my family and I), accepted unanimously to go with DIAGNOS in this adventure, we truly believe both of us will do great and together will achieve important objectives”, declares Quim. Sergio Galindo, founder and owner of Axmilab. “Mr. Andre Larente visited me in Monterrey city, my hometown at my brand new office, we clicked right away because we both are men of one vision: to provide the best diagnostic technology to prevent and leverage the health quality of people.” “I also met Mr. Georges Herbert Chairman of DIAGNOS Inc, during his visit to Mexico City. Very important reasons for me to partner with DIAGNOS, because I partner with true people”, finished Quim. Sergio Galindo.
“We express our gratitude and celebrate along with Axmilab family their decision to collaborate with us, the first step is to prepare ourselves for key projects in different states of Mexico”, mentions Mr. Guillermo Moreno, Vice President of DIAGNOS, Latin America.
About AXIMILAB
Axmilab is a Mexican company with more than 24 years of experience by manufacturing and distributing products in the health sector. His founder, Quim. Sergio Galindo, has successfully driven the business since its conception in 1994, now with the support of his family, is growing exponentially as an example, they’ve been a supplier for IMSS (by winning contracts in the last 13 consecutive years in the diabetes area). Its dominant territory is Northern Mexico, headquarters is in Monterrey, N.L., also covering Mexico City, Sonora, Queretaro, Chiapas, Chihuahua, Durango, Jalisco, Yucatan, San Luis Potosi States among others. Since 1998, Axmilab has been working at all levels of Government, also represents major brands such as Roche, J&J, Licon, PKL, Alifax.
About DIAGNOS
DIAGNOS is a publicly-traded Canadian corporation with a mission of early detection of critical health issues through the use of its Artificial Intelligence (“AI”) tool CARA (Computer Assisted Retina Analysis). CARA is a tele-ophthalmology platform that integrates with existing equipment (hardware and software) and processes at the point of care. CARA’s Artificial Intelligence image enhancement algorithms make standard retinal images sharper, clearer and easier to read. CARA is accessible securely over the internet, and is compatible with all recognized image formats and brands of fundus cameras, and is EMR compatible. CARA is a cost-effective tool for screening large numbers of patients in real-time and has been cleared for commercialization by several regulatory authorities such as Health Canada, the U.S. Food and Drug Administration and the European Union.
Additional information is available at www.diagnos.com and www.sedar.com
For further information, please contact:
| Mr. André Larente, President | Josh Falle |
| DIAGNOS Inc. | Momentum PR |
| Tel: 450-678-8882 ext. 224 | Tel: 514-416-4656 |
| alarente@diagnos.ca | josh@momentumpr.com |
- Published in Diagnos, Life Sciences, News Home, Technology
AREV Brands International Ltd. Announces Asset Purchase Agreement Completed to Bare Topicals from Alternative Extracts Inc.
Momentum Public Relations
Press Release: November 9, 2018
AREV Brands International Ltd. (” AREV ” or the “Company”) ( CSE – AREV ), is pleased to announce it has completed the asset purchase (the “Acquisition Agreement”) dated November 9, 2018 with Alternative Extracts Inc. (“AEI”). To acquire 100% of the Bare Topicals assets in consideration for issuance of 500,000 common shares of the Company at a deemed price of $0.32 per share issued by the Company, for an aggregate value of $160,000.
Bare Brands is a Line of Award Winning Cannabis Infused Topical Products aim to provides safe alternatives to achieving significant pain relief and treatment of skin conditions.
2016
Bio Cup – 1st place topical (Magik)
Bio Cup – 2nd Place topical (SPF 420)
Bio Cup – 1st place topical people’s choice (Magik)
Bio Cup – 2nd Place topical people’s choice (SPF 420)
Bio Cup – 2nd Place best product (overall for the event) (SPF 420)
Karma Cup – 3rd place topical (Magik)
2017
Okanagan Cannabis Cup – 2nd place topical (Magik)
High Times Cannabis Cup – 2nd Place topical (Magik)
Karma Cup – 1st place topical (Recover)
Karma Cup – 3rd place topical (Magik)
2018
The Canna Cup – 1st place topical
The Grey Area Festival – 2nd Place topical (Releaf)
Okanagan Cannabis Cup – 2nd Place topical (Recover)
Karma Cup – 2nd Place topical (Recover)
The most awarded topical company in Canada.
Mike Withrow, CEO for AREV Brands International states “This acquisition is another that adds to the portfolio of products AREV continues to assemble while working on obtaining ACMPR Licencing to sell any of our cannabis based products. In the meantime we are preparing the Bare Topicals herbal line for production.”
The Company, effective immediately, has issued 500,000 stock options to Directors, Officers and Consultants of the Company at a price of $0.41 for a period of 5 years from the issuance date. These options will vest immediately.
Seek safe harbor and forward-looking statements.
For further information, contact Mike Withrow, mike.withrow.vn@gmail.com.
On behalf of the Board,
Mike Withrow
CEO & Director
About AREV Brands International Ltd.
AREV Brands International Ltd. (“AREV”) produces and delivers functional compounds and ingredients from its world-class extraction systems. AREV is revolutionizing the current delivery method of terpenes, cannabinoids and flavonoids. These premium ingredients and formulations are used in products targeted for sale in the natural health, medical, functional food, nutraceutical, sport nutrition and bioceutical markets. AREV innovates through extraction to produce extracts from specific selected plant and exude from trees that address 5 areas of health including Anxiety, Pain Management, Insomnia, Central Nervous System Disorders & Libido.
- Published in AREV Nutrition Sciences, Business, Life Sciences, Medical Marijuana, News Home, Technology
DIAGNOS Announces Private Placement of Common Stocks and Stock Warrants
Momentum Public Relations
Press Release: November 6, 2018
DIAGNOS Inc. (“DIAGNOS” or the “Corporation”) (TSX Venture: ADK) (OTCQB: DGNOF), a leader in early detection of critical health issues through the use of Artificial Intelligence, announces a private placement (“Private Placement”) of up to 16,160,000 units (each a “Unit”) issued at $0.05 per Unit for gross proceeds of up to $808,000. Each Unit consists of;
- one common share (“Share”), and
- one stock warrant (“Warrant”) entitling the holder to purchase one Share per Warrant at a price of $0.05 per Share, for a period of 24 months from the date of issuance of the Warrant.
The proceeds will be used mainly to fund sales and marketing as well as administrative expenses.
It is expected that one participant in the Private Placement will become a “related party” of DIAGNOS within the meaning of Multilateral Instrument 61-101 – Protection of Minority Security Holders in Special Transactions (“MI 61-101”). It is expected that 15,000,000 Units will be subscribed by the participant. The participant will exercise, as a result of the Private Placement, control up to 19.10% of the common shares of DIAGNOS. The transaction is exempt from the valuation requirement and the minority approval requirement prescribed in MI 61-101 based on the fact that the fair market value of the related party’s participation in the Private Placement does not exceed 25% of DIAGNOS’ market capitalization prior to the closing of the Private Placement.
Shares issued as part of the Private Placement as well as the underlying Shares to be issued upon exercise of the Warrants are subject to a statutory four-month hold period from the date of issuance.
Closing of the Private Placement is scheduled for November 9, 2018. The Private Placement is subject to receipt of all required regulatory approvals, including the approval of the TSX Venture Exchange, as well as the execution of formal documentation.
All monies quoted in this press release shall be stated and paid in lawful money of Canada.
About DIAGNOS
DIAGNOS is a publicly-traded Canadian corporation with a mission of early detection of critical health issues through the use of its Artificial Intelligence (“AI”) tool CARA (Computer Assisted Retina Analysis). CARA is a tele-ophthalmology platform that integrates with existing equipment (hardware and software) and processes at the point of care. CARA’s Artificial Intelligence image enhancement algorithms make standard retinal images sharper, clearer and easier to read. CARA is accessible securely over the internet, and is compatible with all recognized image formats and brands of fundus cameras, and is EMR compatible. CARA is a cost-effective tool for screening large numbers of patients in real-time and has been cleared for commercialization by several regulatory authorities such as Health Canada, the U.S. Food and Drug Administration and the European Union.
Additional information is available at www.diagnos.com and www.sedar.com.
For further information, please contact:
| Mr. André Larente, President | Josh Falle | ||
| DIAGNOS Inc. | Momentum PR | ||
| Tel: 450-678-8882 ext. 224 | Tel: 514-416-4656 | ||
| alarente@diagnos.ca | josh@momentumpr.com | ||
- Published in Business, Diagnos, Life Sciences, News Home, Technology