ATW Tech Provides Additional Information Regarding Signature of a Letter of Intent for Major Acquisition of Greywolf Entertainment Group
Momentum Public Relations
Press Release: November 16, 2018
ATW Tech Inc. (“ATW Tech”) (TSX-V: ATW) has, as previously announced on November 6, 2018, signed a letter of intent for the acquisition from Fastrack Trust (the “Trust”) of all of the outstanding shares of 6855318 Canada Ltd. and 6890202 Canada Ltd. (“Greywolf Entertainment Group” or “Greywolf”). For the unaudited financial year ended December 31, 2017, Greywolf’s revenues were $10.0 million ($11.8 million in 2016) for a net income of $196k ($350k in 2016), with total assets of $2.5 million ($2.4 million) and negative shareholder’ equity of $914k ($759k).
Greywolf Entertainment Group, based in Calgary, provides turnkey full commercial production for interactive TV and/or voting networks required for such shows for landline and mobile consumers. Greywolf’s subsidiaries include Triton Global Business Services Inc. (“Triton”) and Fastrack Global Billing Networks Inc. (“Fastrack”). Triton is a North American clearinghouse that provide billing and networks for phone bills in North America and over 40 other countries. Triton has been listed in the top 300 technology companies from Canada several times. Fastrack is a North American inter-exchange carrier providing networks to its switch centers that intelligently routes consumer traffic to optimized billing or response solutions. Its network is designed to handle mass calling for both short and long periods of time, using its own robust backbone Standard Internet Protocol (SIP) networks to transport traffic anywhere in the world.
The letter of intent contemplates a purchase price of $2.95 million, which is subject to certain adjustments and payable through a cash payment of $2.0 million at closing, a vendor take-back note of $0.3 million payable in 30 equal monthly installments and $0.65 million worth of common shares of ATW Tech. No commission is payable and no change of control will result.
“We are proud to come to an agreement on this strategic acquisition of Greywolf that will help us consolidate our position in the Canadian carrier billing market, accelerate our growth, while almost doubling our revenues, and benefiting from significant cost synergies. Along with the divestiture of the Atman division completed on October 31, 2018, which represented 9.5% of ATW Tech’s revenues in 2017, this contemplated acquisition reflects our will and strategy to concentrate our activities in the financial technologies (“fintech”), particularly on our core integrated payment, interactive communication and voting platforms. This acquisition shall be the first step of our acquisition growth plan with an international reach expected for 2019” said Michel Guay, President and CEO of ATW Tech.
The closing of this transaction between ATW Tech and Greywolf, which are dealing at arm’s length, is conditional among other things on ATW Tech carrying out a satisfactory due diligence on Greywolf, obtaining satisfactory financing and all necessary regulatory approvals (including that of the TSX Venture Exchange). Closing of this transaction is expected before the end of 2018.
The Company also announces that a total of 900 000 share purchase options have been granted to a consultant of the Company at a strike price of $0.10 pursuant to the terms of its share option plan (the “Plan”) and expiring on November 15, 2023.
- Published in Atmanco, ATWTECH, Business, Financial Technology, News Home, Technology
AREV Brands International Ltd. announces Success in Terpene Blends on 1st Degree Burns and Insect Bites
Momentum Public Relations
Press Release: November 14, 2018
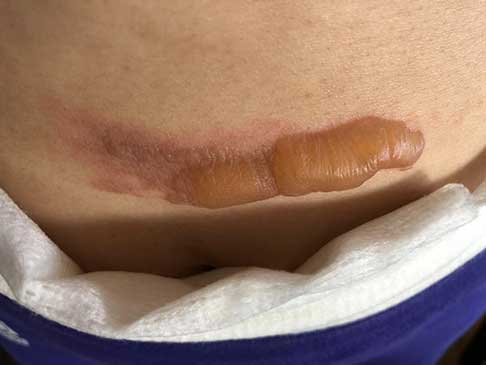
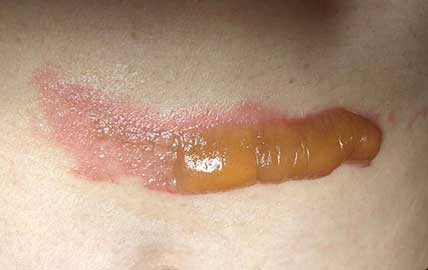
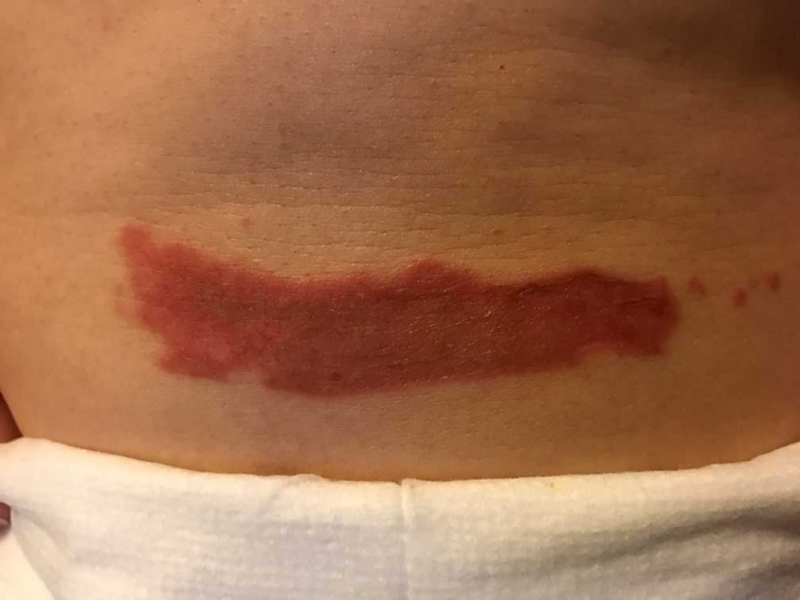
AREV Brands International Ltd. (” AREV ” or the “Company”) ( CSE – AREV ), is pleased to announce AREV Brands International Ltd. has has reached another major milestone in one of its several joint development projects with Alternative Extracts Inc. Further to the research work reported on September 20, 2018, terpenes from non-regulated sources were used to formulate a proprietary composition for burns and insect bites. As noted in peer-reviewed literature, terpene synthesis is vital for cannabinoid synthesis as it provides the GPP molecule for CBGA synthesis.
As evidenced in these images (which was first taken within 18 hours of the burn accident, and in 24-hour succession thereafter), the blisters from the burn were completely eliminated within 4 days from the first application. The patient in this case is a 50-year old woman who has a known history of keloid formation. It is noted that the patient did not develop any infection.
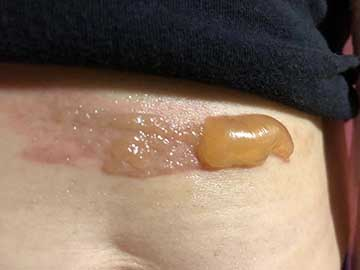
AREV extracted the valuable terpenes from selected non-regulated plant materials and exudates from trees using its extraction systems that it acquired from Alternative Extracts Inc. The composition was formulated in collaboration between AREV and AEI. The mechanism of the composition is believed to be the result of the poly-molecular function of the terpenes.
The company cautions, that although these results are encouraging, a wider sample population is required. A formal study will be conducted in the coming months.
Burn data from World Health Organization shows that:
- – In India, over 1,000,000 people are moderately or severely burnt every year.
– Nearly 173,000 Bangladeshi children are moderately or severely burnt every year.
– In Bangladesh, Colombia, Egypt and Pakistan, 17% of children with burns have a temporary disability and 18% have a permanent disability.
– Burns are the second most common injury in rural Nepal, accounting for 5% of disabilities.
– In 2008, over 410,000 burn injuries occurred in the United States of America, with approximately 40,000 requiring hospitalization.
Mike Withrow commented “Although we are pleased to have been able to minimize the pain and scarring in this burn patient, the results from the use of our proprietary blend of natural compounds warrants further testing and trials.”
Dr. Adrian Wade, Chief Science Officer, of Alternative Extracts Inc. commented, “This collaboration continues to produce highly effective compounds and formulations. Terpenes have demonstrated to play an important role in an increasing number of natural health and medical products.”
Seek safe harbor and forward looking statements.
For further information, contact Mike Withrow, mike.withrow.vn@gmail.com, (778) 896-6536?.
On behalf of the Board,
Mike Withrow
CEO & Director
About AREV Brands International Ltd.
AREV Brands International Ltd. (“AREV”) produces and delivers functional compounds and ingredients from its world-class extraction systems. AREV is revolutionizing the current delivery method of terpenes, cannabinoids and flavonoids. These premium ingredients and formulations are used in products targeted for sale in the natural health, medical, functional food, nutraceutical, sport nutrition and bioceutical markets. AREV innovates through extraction to produce extracts from specific selected plant and exude from trees that address 5 areas of health including Anxiety, Pain Management, Insomnia, Central Nervous System Disorders & Libido.
- Published in AREV Nutrition Sciences, Business, Life Sciences, Medical Marijuana, News Home, Technology
AREV Brands International Ltd. Announces Asset Purchase Agreement Completed to Bare Topicals from Alternative Extracts Inc.
Momentum Public Relations
Press Release: November 9, 2018
AREV Brands International Ltd. (” AREV ” or the “Company”) ( CSE – AREV ), is pleased to announce it has completed the asset purchase (the “Acquisition Agreement”) dated November 9, 2018 with Alternative Extracts Inc. (“AEI”). To acquire 100% of the Bare Topicals assets in consideration for issuance of 500,000 common shares of the Company at a deemed price of $0.32 per share issued by the Company, for an aggregate value of $160,000.
Bare Brands is a Line of Award Winning Cannabis Infused Topical Products aim to provides safe alternatives to achieving significant pain relief and treatment of skin conditions.
2016
Bio Cup – 1st place topical (Magik)
Bio Cup – 2nd Place topical (SPF 420)
Bio Cup – 1st place topical people’s choice (Magik)
Bio Cup – 2nd Place topical people’s choice (SPF 420)
Bio Cup – 2nd Place best product (overall for the event) (SPF 420)
Karma Cup – 3rd place topical (Magik)
2017
Okanagan Cannabis Cup – 2nd place topical (Magik)
High Times Cannabis Cup – 2nd Place topical (Magik)
Karma Cup – 1st place topical (Recover)
Karma Cup – 3rd place topical (Magik)
2018
The Canna Cup – 1st place topical
The Grey Area Festival – 2nd Place topical (Releaf)
Okanagan Cannabis Cup – 2nd Place topical (Recover)
Karma Cup – 2nd Place topical (Recover)
The most awarded topical company in Canada.
Mike Withrow, CEO for AREV Brands International states “This acquisition is another that adds to the portfolio of products AREV continues to assemble while working on obtaining ACMPR Licencing to sell any of our cannabis based products. In the meantime we are preparing the Bare Topicals herbal line for production.”
The Company, effective immediately, has issued 500,000 stock options to Directors, Officers and Consultants of the Company at a price of $0.41 for a period of 5 years from the issuance date. These options will vest immediately.
Seek safe harbor and forward-looking statements.
For further information, contact Mike Withrow, mike.withrow.vn@gmail.com.
On behalf of the Board,
Mike Withrow
CEO & Director
About AREV Brands International Ltd.
AREV Brands International Ltd. (“AREV”) produces and delivers functional compounds and ingredients from its world-class extraction systems. AREV is revolutionizing the current delivery method of terpenes, cannabinoids and flavonoids. These premium ingredients and formulations are used in products targeted for sale in the natural health, medical, functional food, nutraceutical, sport nutrition and bioceutical markets. AREV innovates through extraction to produce extracts from specific selected plant and exude from trees that address 5 areas of health including Anxiety, Pain Management, Insomnia, Central Nervous System Disorders & Libido.
- Published in AREV Nutrition Sciences, Business, Life Sciences, Medical Marijuana, News Home, Technology
CROP Working ‘Rapidly’ To Vertically Integrate California Production, Extraction, Distribution And Retail
Momentum Public Relations
Press Release: November 7, 2018
CROP INFRASTRUCTURE CORP. (CSE: CROP) (OTC: CRXPF) (Frankfurt: 2FR) announced today that it is working towards complete vertical integration in California. Processing continues at Humboldt Farm and new automation equipment has arrived to increase efficiencies and continually increase return on investment (ROI) of finished inventory.
Additionally, CROP’s Emerald Heights retail brand has just completed the stage three interview process with the City of San Bernardino which is a major hurdle before the final licensing review to open its first California retail location. CROP is currently going through the process of opening two Emerald Heights locations in Italy, one in Nevada and one at the aforementioned location in California.
The company’s tenant is currently accepting and reviewing bids from distributors to represent the company’s production under its Hempire, Evolution and White Rhino brands. The tenant has also applied for its own distribution license to represent its own production and the production of other complimentary producers in the region which will result in another license in the growing portfolio of tenant licensees.
CROP has also been notified that the tenants are preparing an extraction license application for Humboldt Farm which will maximize the ROI and broaden the range of Stock Keeping Units (‘SKUs’) available to retail locations.
CROP has submitted its building plans to the Humboldt County Building and Planning Department to increase the production of the California facility at a cost of $1,000,000 of which $250,000 has already been spent. The increased production will result in an additional ~12,000 pounds of high-quality cannabis and 3,000 pounds of secondary material per year.
CROP Infrastructure CEO, Michael Yorke, stated: “The significance of fully vertically integrating cannot be understated and we are working towards that end as rapidly as is practicable. As with CROP’s worldwide tenant strategy, production is always focused on high quality at low cost. With extraction and retail verticals now in process, the opportunity to maximize ROI on a significant scale presents the opportunity to control CROP’s tenant destiny and maximize future profits.”
About CROP
Crop Infrastructure Corp. is publicly listed on the Canadian Securities Exchange and trades under the symbol “CROP” and in the US under the symbol “CRXPF”. CROP is primarily engaged in the business of investing, constructing, owning and leasing greenhouse projects as part of the provision of turnkey real estate solutions for lease-to-licensed cannabis producers and processors offering best-in-class operations. The Company’s portfolio of projects includes cultivation properties in California, two in Washington State, a 1,000-acre Nevada Cannabis farm, an 1,865 acre CBD farm, extraction in Nevada with international focuses in Jamaica and Italy and a joint venture on West Hollywood and San Bernardino dispensary applications.
CROP has developed a portfolio of assets including Canna Drink, a cannabis infused functional beverage, US and Italian distribution rights to over 55 cannabis topical products and a portfolio of 16 Cannabis brands.
- Published in Business, CROP Infrastructure, Marijuana, Medical Marijuana, News Home
DIAGNOS Announces Private Placement of Common Stocks and Stock Warrants
Momentum Public Relations
Press Release: November 6, 2018
DIAGNOS Inc. (“DIAGNOS” or the “Corporation”) (TSX Venture: ADK) (OTCQB: DGNOF), a leader in early detection of critical health issues through the use of Artificial Intelligence, announces a private placement (“Private Placement”) of up to 16,160,000 units (each a “Unit”) issued at $0.05 per Unit for gross proceeds of up to $808,000. Each Unit consists of;
- one common share (“Share”), and
- one stock warrant (“Warrant”) entitling the holder to purchase one Share per Warrant at a price of $0.05 per Share, for a period of 24 months from the date of issuance of the Warrant.
The proceeds will be used mainly to fund sales and marketing as well as administrative expenses.
It is expected that one participant in the Private Placement will become a “related party” of DIAGNOS within the meaning of Multilateral Instrument 61-101 – Protection of Minority Security Holders in Special Transactions (“MI 61-101”). It is expected that 15,000,000 Units will be subscribed by the participant. The participant will exercise, as a result of the Private Placement, control up to 19.10% of the common shares of DIAGNOS. The transaction is exempt from the valuation requirement and the minority approval requirement prescribed in MI 61-101 based on the fact that the fair market value of the related party’s participation in the Private Placement does not exceed 25% of DIAGNOS’ market capitalization prior to the closing of the Private Placement.
Shares issued as part of the Private Placement as well as the underlying Shares to be issued upon exercise of the Warrants are subject to a statutory four-month hold period from the date of issuance.
Closing of the Private Placement is scheduled for November 9, 2018. The Private Placement is subject to receipt of all required regulatory approvals, including the approval of the TSX Venture Exchange, as well as the execution of formal documentation.
All monies quoted in this press release shall be stated and paid in lawful money of Canada.
About DIAGNOS
DIAGNOS is a publicly-traded Canadian corporation with a mission of early detection of critical health issues through the use of its Artificial Intelligence (“AI”) tool CARA (Computer Assisted Retina Analysis). CARA is a tele-ophthalmology platform that integrates with existing equipment (hardware and software) and processes at the point of care. CARA’s Artificial Intelligence image enhancement algorithms make standard retinal images sharper, clearer and easier to read. CARA is accessible securely over the internet, and is compatible with all recognized image formats and brands of fundus cameras, and is EMR compatible. CARA is a cost-effective tool for screening large numbers of patients in real-time and has been cleared for commercialization by several regulatory authorities such as Health Canada, the U.S. Food and Drug Administration and the European Union.
Additional information is available at www.diagnos.com and www.sedar.com.
For further information, please contact:
| Mr. André Larente, President | Josh Falle | ||
| DIAGNOS Inc. | Momentum PR | ||
| Tel: 450-678-8882 ext. 224 | Tel: 514-416-4656 | ||
| alarente@diagnos.ca | josh@momentumpr.com | ||
- Published in Business, Diagnos, Life Sciences, News Home, Technology
Canada Cobalt Drills 2.28% Cobalt Over 7 Meters at Castle
Momentum Public Relations
Press Release: November 2, 2018
Canada Cobalt Works Inc. (TSXV: CCW) (OTC: CCWOF) (Frankfurt: 4T9B) (the “Company” or “Canada Cobalt”) is pleased to announce that underground drilling on the first level of the Castle mine, specifically targeting cobalt for the first time at this historic silver producer, has returned high-grade cobalt, nickel and silver grades.
Highlights from the first three drill holes are as follows:
- 2.28% cobalt, 261 g/t silver and 1.65% nickel over 7.00 meters in hole CA18-001
- 1.87% cobalt, 4,763 g/t silver, 1.29% nickel and 1.19 g/t gold over 2.54 m in CA18-002
- 3.16% cobalt and 10,741 g/t silver (345 ounces per tonne) over 0.60 meter in hole CA18-003
Frank Basa, Canada Cobalt President and CEO, commented: “These cobalt grades are very high in a global context and demonstrate the unique opportunity at the Castle mine, from which we have already created battery grade cobalt sulphate through our proprietary Re-2OX process for evaluation by clients in Asia and Europe.
“The purpose of this initial and continuing underground drill program is to confirm that the Castle vein structures do contain impressive cobalt values. Previous operators focused exclusively on mining high-grade silver through the 11 levels, ignoring cobalt and other metals such as nickel and gold.
“We have a lot of room on the first level while pump testing continues to the second level, beginning at a depth of 25 meters. In light of these initial results, we are accelerating efforts at deploying the most effective underground techniques to fully leverage the Castle mine cobalt opportunity for shareholders,” Basa concluded.
Holes CA18-001, 002 & 003
The first three holes targeted a vein structure near the adit entrance and attempted to follow the vein from a series of inclinations from approximately the same drill set-up through the Nipissing Diabase toward the second level in order to test grade potential (follow-up drilling across the structure will assess true widths). Drilling in these holes exited the vein at depths of 7 meters, 6 meters and 9.25 meters, respectively, reaching a maximum hole length of 30 meters, underscoring the potential to identify additional high-grade mineralization at significantly deeper levels through additional drilling in this area and elsewhere. Intervals reported below are core lengths with true thickness unknown at this time.
|
From (m) |
To (m) |
Interval |
Grade Co (%) |
Grade Ag (g/t) |
Grade Ni (%) |
Grade Au (g/t) |
|
|
CA18-001 |
0.00 |
7.00 |
7.00 |
2.28 |
261.44 |
1.65 |
|
|
including |
0.00 |
2.20 |
2.20 |
3.74 |
743.04 |
2.97 |
|
|
including |
4.30 |
6.06 |
1.76 |
2.27 |
50.82 |
2.19 |
|
|
CA18-002 |
2.17 |
6.02 |
3.85 |
1.33 |
3736.50 |
0.92 |
|
|
including |
3.48 |
6.02 |
2.54 |
1.87 |
4763.26 |
1.29 |
1.19 |
|
CA18-003 |
5.02 |
9.25 |
4.23 |
0.46 |
1566.10 |
n/a |
|
|
including |
6.35 |
6.95 |
0.60 |
3.16 |
10741.28 |
0.48 |
The Company eagerly anticipates additional assay results and has fully winterized the Castle mine for continued underground work including drilling over the coming months.
Canada Cobalt also expects to provide an update shortly on the surface drill program east of the mine where a potential new discovery has been made with the third drill hole of the 2018 program (see October 24, 2018, news release).
Quality Assurance/Quality Control
Canada Cobalt Works’ AQTK drill program employs diligent standards in drill core sampling and quality assurance/quality control. Core from the above holes was sent to Swastika Laboratories in Swastika, Ontario, for analysis. Where silver was visually and significantly present, a Pulp-Metallic analysis was completed for the silver assays where the entire sample was dried, weighed and crushed over 95% then fully pulverized and passed through 147-micron screen to create a plus 147-micron fraction (metallics) and a minus 147-micron fraction (pulp). Approximately 50 grams of the metallic fraction was weighed, fused and assayed by Fire Assay. The resulting dore bead of gold and silver was weighed before adding HNO3 acid to dissolve the silver. The remaining gold bead was weighed and subtracted from the gold + silver bead to give a silver weight and grade for the metallics portion. The minus 147-micron fraction (pulp) had two samples from entire pulp which were weighed then digested by two acids aqua-regia and finalized by AAS reading for Co, Ag and Ni. Final silver grade is a calculated weighted average using grades and weights from both pulps and metallics.
As gold was not assayed in the pulps, the final gold assay was calculated by weighted average, as for the silver, but assuming a 0.0 g/t grade for the pulp portion. Cobalt and nickel were reported as percent grade. Other samples were assayed for cobalt, silver and nickel by AAS after aqua-regia digestion. Analytical accuracy and precision are monitored by the analysis of reagent blanks and reference materials at the lab. Quality control is further assured by the insertion of blind certified standard reference material and blanks into the sample stream at regular intervals by Canada Cobalt Works personnel in order to independently assess analytical accuracy.
Qualified Person
The technical information in this news release was prepared under the supervision of Frank J. Basa, P.Eng., Canada Cobalt’s President and Chief Executive Officer, who is a member of Professional Engineers Ontario and a qualified person in accordance with National Instrument 43-101.
About Canada Cobalt Works Inc.
Canada Cobalt is a pure play cobalt company focused on its past producing Castle mine in the Northern Ontario Cobalt Camp, Canada’s most prolific cobalt district. With underground access at Castle, a recently installed pilot plant to produce cobalt-rich gravity concentrates on site, and a proprietary hydrometallurgical process known as Re-2OX for the creation of technical grade cobalt sulphate as well as nickel-manganese-cobalt (NMC) formulations, Canada Cobalt is strategically positioned to become a vertically integrated North American leader in cobalt extraction and recovery.
“Frank J. Basa”
Frank J. Basa, P. Eng.
President and Chief Executive Officer
Neither the TSX Venture Exchange nor its Regulation Service Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release. This news release may contain forward-looking statements including but not limited to comments regarding the timing and content of upcoming work programs, geological interpretations, receipt of property titles, potential mineral recovery processes, etc. Forward-looking statements address future events and conditions and therefore, involve inherent risks and uncertainties. Actual results may differ materially from those currently anticipated in such statements.
SOURCE Canada Cobalt Works Inc.
- Published in Business, Canada Cobalt Works, Mining, News Home
ATW Tech Closes the Sale of its Atman Assets for $1.3 Million
Momentum Public Relations
Press Release: November 1, 2018
ATW Tech Inc. (TSX-V: ATW) announces today the closing of the sale of its Psychometric Research and Services of Atman assets (“Atman”), previously announced on September 17, 2018. Those assets consist primarily of the personality test platforms and learning modes offered to businesses for human resources management. The total purchase price is $1,300,000, composed of $1,100,000 paid immediately in cash and $200,000 as monthly installments over the next 3 years and based on a percentage of sales. ATW Tech will pay to the buyer a total of $95,000 in twelve equal monthly instalments for its working capital. For the fiscal year ended December 31, 2017, revenues generated by the Atman assets being sold represented 9.6% of ATW Tech’s revenues.
“We are proud to conclude this transaction, and especially to do so within the expected timeline. This transaction reflects our will and strategy to concentrate all our activities and resources on our financial technologies (“fintech”)” said ATW Tech’s President and CEO, Michel Guay.
Additional information regarding the Company is available on SEDAR www.sedar.com. The TSX Venture Exchange and its Regulatory Services provider (as per meaning assigned to this term in TSX Venture Exchange’s policies) bear no liability as to the relevance or accuracy of this press release.
ABOUT ATW TECH
ATW Tech (TSX-V: ATW) is a leader in information technology, owner of several web platforms including VoxTel, VuduMobile, Option.vote and Bloomed. VoxTel offers various interactive communication and landline and mobile carrier billing phone solutions. VuduMobile is specialized the text messaging business for enterprises through its unique, user-friendly and bilingual test messaging application et turnkey solution allowing management of text message management programs in all kind of businesses. Option.vote offers a large scale customizable and secured multi-method voting system for unions, political parties, professional associations, and others looking for a way to reduce their voting costs and to improve their participation rates. Bloomed is a cloud-based platform to manage data (smart data) on consumers and their behaviors which is developed for marketing agencies and their campaigns for the consumer and corporate markets.
SOURCE:
| ATW Tech Michel Guay Founder, president and CEO Tel.: 514.935.5959 ext. 301 mguay@atwtech.com www.atwtech.com |
Simon Bédard, CA, CPA, CFA, MBA CFO Tel. : 514.935.5959 ext. 304 sbedard@atmanco.com |
- Published in Atmanco, Business, Financial Technology, News Home, Technology
Tetra Bio-Pharma Obtains Health Canada Approval to Conduct a Phase 1 Pharmacokinetic & Safety Study Using Vaporized Version of PPP001
Momentum Public Relations
Press Release: November 1, 2018
Tetra Bio-Pharma Inc. (“Tetra” or “TBP”) today announced that is has received a “No Objection Letter” (NOL) from the Therapeutic Drug Directorate at Health Canada to conduct a Phase 1 pharmacokinetic (PK) and safety study using a vaporized version of PPP001.
This study aims to determine the Pharmacokinetic and safety profile of PPP001 delivered as an inhaled vapour using the Mighty Medic (see previous July 16, 2018 News Release), thus providing patients with an alternative to the smoked version of PPP001 and allowing Tetra to develop PPP001 for healthier patients suffering from uncontrolled pain. It is anticipated that this phase 1 data, combined with Tetra’s characterization of the composition of vapor, will provide the necessary evidence to advance the development of vaporized PPP001 into more chronic and non-life-threatening diseases, such as fibromyalgia. The study is expected to begin and end in Q4 2018.
“We are pleased to announce this critical Phase 1 trial focused on the PK and safety of our PPP001 pellet delivered by vaporization,” said Dr. Guy Chamberland, CEO and CSO of Tetra Bio-Pharma Inc. “Over the last 3 months we have analyzed the composition of the vapor. This data combined with the results of this Phase 1 will provide the critical pharmacological information required by physicians to adequately guide future efficacy studies in patients who suffer from a chronic condition such as fibromyalgia where there are limited treatment options and there is a large unmet medical need. It may also provide an alternative mode of delivery for those patients who prefer not to smoke.”
About Tetra Bio-Pharma
Tetra Bio-Pharma (TSX-V: TBP) (OTCQB: TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and development with a Health Canada approved, and FDA reviewed, clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. The Company has several subsidiaries engaged in the development of an advanced and growing pipeline of Bio Pharmaceuticals, Natural Health and Veterinary Products containing cannabis and other medicinal plant-based elements. With patients at the core of what we do, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies. For more information visit: www.tetrabiopharma.com
- Published in Business, Life Sciences, Medical Marijuana, News Home, Tetra Bio Pharma
CROP Developing One Ton Nevada Extraction Facility With 60% Payment Coming From 1,500,000 Pound Supply Agreement
Momentum Public Relations
Press Release: November 1, 2018
CROP INFRASTRUCTURE CORP. (CSE: CROP) (OTC: CRXPF) (Frankfurt: 2FR) announced today that the company has now engaged an ISO/GMP rated extraction facility developer to build a one ton per day extraction facility at its Nevada farm.
On October 23rd CROP announced a 500,000 pound annual (1,500,000 over 3 years) supply agreement with a commercial extraction technology company. The same company will be developing, and providing, extraction equipment for the CROP Nevada extraction facility. The initial $3.2 Million, equivalent to ~89,000 pounds of product, will be allocated towards paying for 60% of the extraction facility. The balance of ~$2.0 Million, due in cash over the next eight months will be financed by revenue from the company’s operations.
As previously announced, the CBD flower off take is to be delivered in shipments of 50,000 dry pounds on a bi-monthly basis with payment to be made plus the cost of delivery of $36.00 per pound to $57.00 per pound depending on Certificate of Analysis of the CBD content with deliveries starting August 2019.
Cannabis industry analysts, The Brightfield Group, estimate the hemp-CBD market alone could reach $22-billion by 2022.
CROP is in process of harvesting its 240 acres planted this year, which will be processed for isolate and is also readying its 1,865-acre CBD farm for 2019 planting.
CROP CEO, Michael Yorke, stated: “The synergies and relationship between CROP and our off-take partner continue to evolve. We look forward to continuing to develop this new partnership into new opportunities and other territories. We believe this is an effective and creative way of financing and developing our growth in a non-dilutive arrangement.”
About CROP
Crop Infrastructure Corp. is publicly listed on the Canadian Securities Exchange and trades under the symbol “CROP” and in the US under the symbol “CRXPF”. CROP is primarily engaged in the business of investing, constructing, owning and leasing greenhouse projects as part of the provision of turnkey real estate solutions for lease-to-licensed cannabis producers and processors offering best-in-class operations. The Company’s portfolio of projects includes cultivation properties in California, two in Washington State, a 1,000-acre Nevada Cannabis farm, a 1,865-acre CBD farm, extraction in Nevada with international focuses in Jamaica and Italy and a joint venture on West Hollywood and San Bernardino dispensary applications.
CROP has developed a portfolio of assets including Canna Drink, a cannabis infused functional beverage, US and Italian distribution rights to over 55 cannabis topical products and a portfolio of 16 Cannabis brands.
Company Contact
Michael Yorke – CEO and Director
E-mail: info@cropcorp.com
Website: www.cropcorp.com
Phone: (604) 484-4206
- Published in Business, CROP Infrastructure, Marijuana, Medical Marijuana, News Home
Tetra Natural Health Signs Exclusive Agreement for the Distribution of their Hemp Energy Drink in Canada and the USA
Momentum Public Relations
Press Release: November 1, 2018
Tetra Natural Health Inc., a division of Tetra Bio-Pharma Inc. (“Tetra” or the “Company”) (TSX VENTURE: TBP) (OTCQB: TBPMF), today announced that it has signed a major distribution agreement with Kombucha Baby Brewing Company and its partners for the exclusive distribution of its Hemp Energy Drink in the Provinces of British Columbia, Alberta, Saskatchewan, Manitoba, and Ontario as well as California, Washington, Oregon and Hawaii.
The Hemp Energy Drink provides consumers with a product that contains fewer calories and more natural ingredients than existing energy drinks thus providing a welcome alternative to existing products in what is considered to be an extremely lucrative market.
“We are thrilled in having signed this agreement with Kombucha Baby and its distribution partners providing Tetra Natural Health with access to a significant number of retail outlets in both Canada and the USA,” stated Richard Giguere, CEO of Tetra Natural Health.
“We are very excited to be adding the Hemp Energy Drink to our portfolio of products that will be available to our existing and potential client base,” stated Dan McKenna, President and CEO of Kombucha Baby Brewing Company Inc. “Numerous Canadian and U.S. customers representing thousands of retail outlets have indicated a strong interest in carrying this unique Natural Health Product (NHP).”
AboutTetra Bio-Pharma
Tetra Bio-Pharma (TSX-V: TBP) (OTCQB: TBPMF) is a biopharmaceutical leader in cannabinoid- based drug discovery and development with a Health Canada approved, and FDA reviewed, clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. The Company has several subsidiaries engaged in the development of an advanced and growing pipeline of Bio Pharmaceuticals, Natural Health and Veterinary Products containing cannabis and other medicinal plant-based elements. With patients at the core of what we do, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies.
For more information visit: www.tetrabiopharma.com
- Published in Business, Life Sciences, Medical Marijuana, News Home, Tetra Bio Pharma