Former CEO of Jamaica’s Cannabis Licensing Authority Joins CROP’s Executive Advisory Team
Momentum Public Relations
Press Release: August 23, 2018
CROP Infrastructure Corp. (CSE: CROP) (OTCMKTS: CRXPF) (“CROP” or the “Company”) announced today the appointment of Greg Douglas to the Executive Advisory Board who will have a special focus on CROP’s Jamaica operations.
CROP Infrastructure CEO, Michael Yorke, stated, “Greg can be considered one of the foremost authorities on the Jamaican cannabis industry. He will be of considerable assistance to CROP as the business develops there.
“Greg has the invaluable ability to effectively interact with stakeholders from both the private and public sectors and his knowledge of the industry, both in the Caribbean and North America, is truly extensive and that will be of great benefit to CROP as it expands even further internationally.”
Greg Douglas formally entered the Cannabis space in 2015 when he joined the Bureau of Standards Jamaica as their Management Information Systems (MIS) Manager. Shortly after joining the Bureau he was named as the Project Coordinator for a proposed Medical Cannabis (Ganja) Track and Trace Pilot Project.
Through this Greg was exposed to compliance mechanisms used in Colorado, touring state facilities which increased his understanding and appreciation of the industry while building relationships with Colorado’s compliance agency, along with local businesses.
Greg is now known internally at the Bureau as the ‘Ganja Man’ and externally by stakeholders as the ‘Bureau Man’. During April to November 2017 Greg was seconded to the Cannabis Licensing Authority (CLA) as their CEO. While there he became a part of history issuing the first commercial licenses.
He also sought to streamline internal processes to increase the efficiency of the Authority. He was part of the CLA team that visited Canada where the team met with both Canadian Government entities and officials (Health Canada, MP Bill Blair, Department of Finance), also with resident offices of the Jamaica Tourist Board and JAMPRO.
Greg resumed his duties at the Bureau of Standards in January 2018 and is engaged as a member of the team responsible for the creation of standards within the cannabis industry in Jamaica.
About CROP Infrastructure Corp.
CROP Infrastructure Corp. is publicly listed on the Canadian Securities Exchange and trades under the symbol “CROP” and in the US under the symbol “CRXPF”. CROP is primarily engaged in the business of investing, constructing, owning and leasing greenhouse projects as part of the provision of turnkey real estate solutions for lease-to-licensed cannabis producers and processors offering best-in-class operations. The Company’s portfolio of assets includes cultivation properties in California, Washington State, Nevada, Italy, Jamaica and a joint venture on West Hollywood and San Bernardino dispensary applications. CROP has developed a portfolio of 16 Cannabis brands and has US and Italian distribution rights to a line of over 55 topical cannabis products from The Yield Growth Corp.
Company Contact
Michael Yorke – CEO & Director
E-mail: info@cropcorp.com
Website: www.cropcorp.com
Phone: (604) 484-4206
- Published in Business, CROP Infrastructure, Medical Marijuana, News Home
Equitorial drills 30.53 m of 1.67% Li2O at Cat Lake
Momentum Public Relations
Press Release: August 23, 2018
Equitorial Exploration Corp. (TSX-V: EXX, Frankfurt: EE1, OTCQB: EQTXF) (“Equitorial” or “Company”) is pleased to announce assay results from drill hole CT-18-06 of the Cat Tail Pegmatite from its Phase One drill program carried out in late spring 2018 at its 100%-owned Cat Lake Lithium Property in southeast Manitoba.
The company has applied for a drill permit to carry out Phase Two, a 5,000 meter, drill hole program to further explore the lithium and tantalum potential at depth and along strike and intends to commence drilling in September 2018.
Drill Hole CT-18-06
Drill hole CT-18-06 was collared at the same location as the discovery drill hole, CT-18-02. The discovery drill hole, was positioned to intersect a proposed southerly projection of the Irgon Pegmatite system, a pegmatite that is presently being explored by QMC Quantum Minerals to the west of the company’s claims. The discovery drill hole CT-18-02 encountered 36.19 meters of a spodumene-bearing pegmatite (true width not determined at this time) at a depth of 126.41 meters downhole or 90 meters below the surface. Spodumene is one of the major lithium bearing minerals explored for in pegmatites. The hole was collared approximately 222 meters southeast of the last surface exposure of the Irgon Pegmatite. The Cat Tail Pegmatite has no known surface expression.
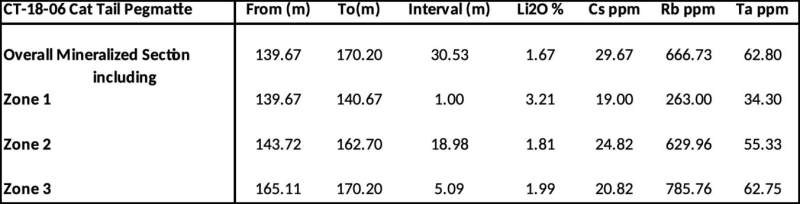
CT-18-06 was collared to intersect the lithium-bearing Cat Tail Pegmatite beneath the discovery hole. It encountered the pegmatite at a depth of 137.47 meters downhole or approximately 120 meters below the surface. The pegmatite encountered in the drill hole was 33.21 meters in core length (true width not determined at this time). Numerous sections of spodumene enrichment in the pegmatite were encountered in the drill core with individual assays up to 3.57% Li 2 O. The overall mineralized section of the pegmatite (see Table 1) graded 1.67 Li 2 O over a drill core length of 30.53 meters (true width not determined at this time) with three zones grading 3.21%, 1.81% and 1.99% (see Table1). The assay results of drill hole CT-18-06 extended the lithium mineralization in the Cat Tail Pegmatite to depth. The pegmatite remains open at depth and along strike.
Table 1: Weighted Averages for drill hole CT-18-06 at the Cat Tail Pegmatite
Summary of the Phase One Cat Tail Pegmatite 2018 Drill Program
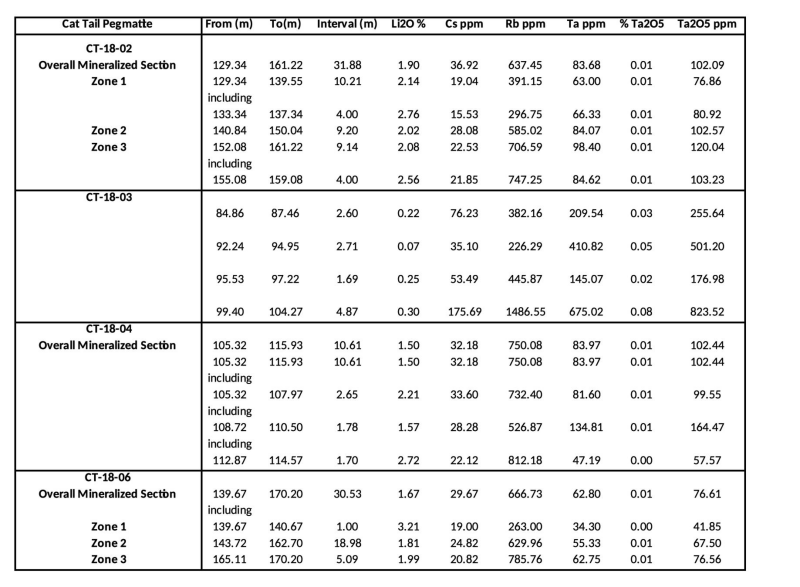
A total of 1,256 meters was completed in the Phase One diamond drill hole program. A total of seven drill holes were drilled with six drill holes testing the lithium and pegmatite potential of the property and the seventh hole testing a sulphide rich skarn. Numerous pegmatites were encountered in all the holes with a main spodumene (lithium mineral) bearing pegmatite (Cat Tail Pegmatite) encountered in four of the six holes. Drill hole CT-18-03 encountered several high grade tantalum zones. Prior to being drilled, this pegmatite was buried with no surface expression. Assay results are still pending for sulphide rich skarn.
Table 2 is an overview of the drill holes that encountered the Cat Tail Pegmatite.
Table 2: Cat Tail Pegmatite Intersections .
Phase Two Drill Program
The company plans to carry out a Phase Two drill hole program on the Cat Tail Pegmatite to further explore the lithium and tantalum potential at depth and along strike. A drill work permit has been applied for with the Province of Manitoba and the company is awaiting approval. The drill program has been designed to further drill test the Cat Tail Pegmatite along strike and at depth. An exploration program of mapping and sampling is scheduled for the fall of 2018. The field work will aid in a better understanding of the Cat Tail Pegmatite system. The company also plans to initiate a PEA (Preliminary Economic Assessment) Study after the completion of the Phase Two drilling.
QA/QC Protocol
All core samples were sent to the Activation Laboratories facility, in Ancaster, Ontario for analysis. Samples were prepared, using the lab’s Code RX1 procedure. Samples are crushed, up to 95% passing through a 10 mesh, riffle split, and then pulverized, with mild steel, to 95%, passing 105 ?m. Analyses were completed, using the lab’s Ultratrace 7 Package; a Sodium Peroxide Fusion which allows for total metal recovery and is effective for analysis of Sulphides and refractory minerals. Assay analyses are carried out, using ICP-OES and ICP-MS instrumentation. Equitorial Exploration implemented a QA/QC program for the drill program with the injection of blanks, duplicates and commercial standards at intervals throughout the sample sequence. Activation Laboratories has their own internal QA/QC procedures that it carries out for all sample batches.
Qualified Person
The drill program was supervised Carey Galeschuk. P. Geo, a consulting geologist with extensive experience in rare metal and lithium-bearing pegmatites. He also serves as Qualified Person for the purpose of National Instrument 43-101.
Cat Lake Lithium Property Highlights
- – Property situated directly east and along strike of Quantum Minerals’ Cat Lake Mineral Project (previously Irgon Lithium Mine) . During 1956-1957, t he Irgon Mine was an underground mining operation for spodumene (one of the hard rock sources for Li). The pegmatite had an historic estimate of 1.25 million tons of ore grading 1.51% Li20 (Mineral Inventory File No. 221).
– The Equitorial Exploration claim block is located 150 m from south end of Irgon Lithium Mine shaft and approximately 93 meter east of the last exposed outcrop of the Irgon Pegmatite
– 48 feet (14.63 m) of spodumene bearing pegmatite were reported drilled in 1948 on the company’s present claims but not followed up at the time (Manitoba Assessment File 98073 – not 43-101 compliant )
– Property approximately 180 km northeast of Winnipeg, Manitoba
– Excellent infrastructure – Provincial Highway 314 in southeast Manitoba cuts through the property
Please click for maps of the claims: http://equitorialexploration.com/wp-content/uploads/2018/01/Cat-Lake-Claims-Maps-3.pdf
Geologically, the property lies within the east-trending Mayville-Cat-Euclid Greenstone Belt (“MCEGB”) located along the northern contact of the Maskwa Lake Batholith. This northern greenstone belt has a similar structural geological setting as the Bird River Greenstone Belt (“BRGB”) which is located along the southern contact of the same batholith, and is parallel to and approximately 18km to the south of the MCEGB. The property is located 20 kilometers north of the Tanco Mine Property. The BRGB hosts the world-class Tanco rare metal-bearing pegmatite as well as numerous other lithium-bearing pegmatites. The Tanco Mine went into production in 1969 and produced tantalum, cesium and spodumene (lithium). It was previously North America’s largest and sole producer of spodumene (Li), tantalum (Ta) and pollucite (Cs).
About Equitorial Exploration Corp
Equitorial is aggressively developing four 100%-owned, high-potential, lithium projects in North America. The Little Nahanni Pegmatite Group (LNPG) is a 43-101 compliant, hard rock, lithium property in the NWT. The Cat Lake Lithium Property in Manitoba, Canada is directly adjacent to the Cat Lake Mineral Project, a highly prospective Lithium property. The Tule and Gerlach Lithium Brine Projects are located in lithium-rich Utah and Nevada within easy reach of the Tesla Gigafactory #1. All four projects have demonstrated highly encouraging grades.
For more information please visit: http://equitorialexploration.com/
On behalf of the Board of Directors
EQUITORIAL EXPLORATION CORP.
_____________________
Jack Bal, CEO and Director
For further information, please contact Jack Bal at 604-306-5285
- Published in Equitorial Exploration, Mining, News Home
Tetra Bio-Pharma applies for orphan drug designation for CBDs
Momentum Public Relations
Press Release: August 23, 2018
Tetra Bio-Pharma Inc.(“Tetra” or “TBP”), today announced the submission of multiple applications for Orphan Drug Designation for cannabinoids to the U.S. Food and Drug Administration, for the treatment of various forms of rare cancers and ocular diseases. The applications align with Tetra’s strategy to develop cannabis prescription drug products for the treatment of several rare cancers and to expand its market exclusivity in the treatment of ocular diseases. If successful, the designation of cannabinoids as an Orphan Drug will open the door for fast track approval, tax credits on clinical research, and reduced registration fees, in addition to other opportunities. It further provides Tetra with a 7-year period of market exclusivity in the U.S.
Today’s announcement builds on previous milestones for Tetra in cancer, ocular disease and pain conditions. In May 2017, Tetra announced its partnership with Panag Pharma to develop innovative cannabinoid-based prescription drugs for the treatment of ocular inflammatory and pain conditions. The company also announced it submitted patent applications to expand its exclusivity for the development of pharmaceutical drug formulations to treat various forms of cancer and ocular disease. This latter protection complements the intellectual property owned by Panag Pharma.
In March 2018, Tetra announced that the U.S. FDA granted an Orphan Drug Designation for Complex Regional Pain Syndrome. The company has expanded its regulatory activities in the USA to accelerate making cannabinoid-based prescription drugs accessible to patients.
“Managing rare forms of cancer presents a huge challenge for physicians,” said Dr. Guy Chamberland, M.Sc., Ph.D., interim CEO and Chief Scientific Officer of Tetra Bio-Pharma. “Our R&D team has submitted numerous Orphan Drug Designation applications to the FDA over the past 6 months, which not only holds promise to improve patient care but lines up with our corporate strategy to develop and commercialize cannabinoid prescription drug products for both rare cancers and ocular diseases in the United States.”
About Tetra Bio-Pharma
Tetra Bio-Pharma (TSX-V: TBP) (OTCQB: TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and development with a Health Canada approved, and FDA reviewed, clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. The Company has several subsidiaries engaged in the development of an advanced and growing pipeline of Bio Pharmaceuticals, Natural Health and Veterinary Products containing cannabis and other medicinal plant-based elements. With patients at the core of what we do, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies.
For more information visit: www.tetrabiopharma.com
Source: Tetra Bio-Pharma
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
- Published in Life Sciences, Medical Marijuana, News Home, Tetra Bio Pharma
Arctic Star Exploration Corp. Enters Option Agreement for Stein Diamond Project Nunavut, Canada
Momentum Public Relations
Press Release: August 23, 2018
Arctic Star Exploration Corp. (TSXV: ADD) (FSE: 82A1) (OTCQB: ASDZF) (“Company”) is pleased to announce that it has entered into an Option Agreement with GGL Resource Corp. (TSXV: GGL) (“GGL”) to earn a 60% interest in Arctic Star’s wholly owned Stein Diamond Project, Nunavut Canada.
The Stein Diamond property consists of 4 contiguous prospecting permits covering an area of 1,065 square kilometers on the Southern Boothia Peninsula, 45 kilometers from tide water. It is located 85 kilometers northwest of the community of Taloyoak, Nunavut which is serviced daily by commercial flights and seasonally by barge.
Stein is an advanced diamond exploration project having the benefit of numerous successive exploration campaigns and over $1.5 million in previous exploration expenditures. This historic work included multiple seasons of heavy mineral sampling in which kimberlitic indicator minerals were followed in the up-ice direction to a region believed to be the source area. This area was subsequently flown with detailed airborne magnetic surveys. No kimberlite drill testing has been conducted on the project to date.
The indicator mineral suite contains grains that are indicative of diamond inclusion chemistry showing high chrome, low calcium G10D pyrope garnets. The detailed airborne magnetic surveys have identified numerous high priority targets that have signatures similar in characteristics to kimberlites found elsewhere in Canada’s north. The Stein project is further complimented with the existence of a major structural feature identified on regional government airborne magnetic surveys which is greater than 100 kilometers in length and traverses the area of the project containing the high priority targets. In fields elsewhere, kimberlites can be geologically observed exploiting larger structural features and utilizing them as conduits for emplacement.
The nearest known kimberlite discovery is over 230 kilometers to the southeast and perpendicular the regional ice flow direction. The distance and direction greatly reduces the potential for the Stein mineral grains being an overprint from this field.
Historically, the Stein project was identified and advanced by previous explorers from 2004 to 2008 with a small amount of additional work in 2011. The Global Financial Crisis in 2008 and resulting inability to fund exploration programs essentially halted significant advancement of the project as the next step was to drill test the compelling geophysical targets with associated supporting geochemistry. The lack of advancement caused the mineral tenure to lapse. Arctic Star reacted to this opportunity and acquired the mineral rights in 2015. Arctic Star’s recent focus and deployment of resources has been the ongoing successful exploration of the Timantti Diamond Project in Finland, leaving Stein in portfolio yet to be advanced to drill testing.
GGL can acquire a 60% undivided interest in the Stein Project by conducting detailed ground geophysics on high priority airborne targets and discovering kimberlite by drilling, trenching or in outcrop. Once kimberlite is discovered, a Joint Venture is triggered with an initial 60/40 contributing relationship.
The Company is pleased to be partnered with Arctic Star on this compelling diamond project which is on the cusp of potentially delivering a new, yet undiscovered Canadian kimberlite field. The project has a Class A land use permit which includes drilling.
Nunavut represents a favorable mining and exploration jurisdiction both globally and within Canada. Canadian junior diamond explorers such as Peregrine Diamonds Ltd. (Chidliak Project), Dunnedin Ventures Inc. (Kahuna Project) and North Arrow Minerals Inc. (Mel and Nuajaat Projects) have successfully conducted drilling programs in Nunavut during the 2017 — 2018 seasons. Nunavut currently has three operating mines, one of which received operational status as recently as 2017.
The technical information in this news release has been approved by David Kelsch, P.Geo, President of GGL Resources Corp. and a qualified person for the purposes of National Instrument 43-101.
About Arctic Star
The Company owns 100% of the recently acquired Timantti Diamond Project including a 243 Ha Exploration Permit and a 193,700 Ha Exploration Reservation near the town of Kuusamo, in Finland. The project is located approximately 550km SW of the operating Grib Diamond Mine in Russia. Arctic has commenced its exploration in Finland on the Timantti Project, where four diamondiferous kimberlite bodies may represent the first finds in a large kimberlite field. The Company also controls diamond exploration properties in Nunavut (Stein), the NWT (Diagras and Redemption) and a rare metals project in BC (Cap).
Arctic Star has a highly experienced diamond exploration team previously responsible for several world class diamond discoveries.
ON BEHALF OF THE BOARD OF DIRECTORS OF ARCTIC STAR EXPLORATION CORP.
Scott Eldridge, President & CEO
+1 (604) 722-5381
scott@arcticstar.ca
Patrick Power, Executive Chairman
+1 (604) 218-8772
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-Looking Statements: Certain statements in this press release are forward-looking statements within the meaning of applicable securities laws. Forward-looking statements in this press release include that the Timantti Project transaction is a pre-eminent opportunity.
- Published in Arctic Star Exploration, Mining, News Home
CROP’s Nevada Hemp Farm Adds 750 Acres of Production Land for 2019 Season
Momentum Public Relations
Press Release: August 22, 2018
CROP Infrastructure Corp. (CSE: CROP) (OTC: CRXPF) (“CROP“ or the “Company“) announced today its Nevada subsidiary has leased an additional 750 acres of contiguous agricultural farmland bringing the total Nevada acreage to 1,065 acres with 240 acres under pivot.
The lease also provides access to over 300 acres of additional water rights. The additional 750 acres of pivot development potential will substantially increase production for the 2019 season.
The newly-leased acreage comes with four housing units and a building to be converted into an extraction facility to process hemp biomass for CBD isolate on site. The two-year lease will annually cost $566 USD per acre and the company intends to use a portion of the revenue from its first crop to pay for the lease.
The previously announced 240 acres of CBD Hemp was planted and is currently 1.5 feet tall and growing under pivot. All harvesting equipment has been secured for the resulting hemp biomass. It is estimated that the 240 acre pivots will yield 240,000 pounds of hemp flower. The cost of production is not expected to exceed $700,000 USD with many of the costs already incurred. The first harvest is expected in early Q4 this year.
CROP Infrastructure Director and CEO, Michael Yorkestated: “As previously announced in July, our Nevada Hempfarm and licensed tenant became the largest hemp farmer in Nevada in 2018. We are confident with over 1,000 acres that our tenant will remain the largest hemp farm in Nevada. The next phase of development will be a state-of-the-art extraction facility to make high-value CBD isolate.”
About CROP
Crop Infrastructure Corp. is publicly listed on the Canadian Securities Exchange and trades under the symbol “CROP” and in the US under the symbol “CRXPF”. CROP is primarily engaged in the business of investing, constructing, owning and leasing greenhouse projects as part of the provision of turnkey real estate solutions for lease-to-licensed cannabis producers and processors offering best-in-class operations. The Company’s portfolio of projects includes cultivation properties in California and Washington State, Nevada, Italy, Jamaica and a joint venture on West Hollywood and San Bernardino dispensary applications. CROP has developed a portfolio of 16 Cannabis brands and has US and Italian distribution rights to a line of over 55 cannabis topical products from The Yield Growth Corp.
- Published in Business, CROP Infrastructure, Medical Marijuana, News Home
Auxico Closes First Tranche of Private Placement
Momentum Public Relations
Press Release: August 21 2018
Auxico Resources Canada Inc. (CSE: AUAG) is pleased to announce that it has completed the first tranche of a non-brokered private placement of 2,550,000 units (“Units”), issued at a price of $0.20 per Unit, for gross proceeds of $510,000. Each unit consists of one common share (“Common Share”) and one-half transferable common share purchase warrant (“Warrant”). Each full Warrant entitles the holder to acquire one additional Common Share of the Company at a price of $0.40 per Common Share for two years from the date of issuance.
The Company paid finder’s fees of $35,000 in connection with the private placement.
The net proceeds of the private placement will be used for geological work on the Company’s Zamora Property in Mexico and its mining interests in Colombia, and for general working capital. More specifically, Auxico will begin geological work in Colombia to evaluate coltan opportunities there, as per the Company’s news release of July 10, 2018.
The Company intends to close one or more additional tranches of this private placement on the same terms as presented above, for total gross proceeds (in all tranches) of up to $1,500,000.
Mark Billings, President of the Company stated, “We are very pleased with the support that we have received from investors in completing the first tranche of this private placement, and we publicly thank them for their confidence in Auxico. We also intend on closing additional tranches of this private placement, as the summer now draws to a close. Funds from the private placement will enable us to move ahead with our analysis of many interesting mining opportunities in Colombia.”
ON BEHALF OF THE BOARD OF DIRECTORS
| « signed » | « signed » | |
| Pierre Gauthier | Mark Billings | |
| CEO, Auxico Resources Canada Inc. | President, Auxico Resources Canada Inc. | |
| pg@auxicoresources.com | mb@auxicoresources.com | |
| Cell: +1 514 299 0881 | Cell: +1 514 296 1641 |
About Auxico Resources Canada Inc.
Auxico Resources Canada Inc. (“Auxico”) is a Canadian company that was founded in 2014 and based in Montreal. Auxico is engaged in the acquisition, exploration and development of mineral properties in Colombia and Mexico.
The Canadian Securities Exchange (CSE) has not reviewed and does not accept responsibility for the adequacy or the accuracy of the contents of this release.
- Published in Uncategorized
ATW Tech Increases Its Revenues to Reach $3.2m for Q2-2018
Momentum Public Relations
Press Release: August 20 2018
ATW Tech Inc. (the “Company”) (TSX-V: ATW) announces its financial results for its second quarter ended June 30, 2018.
For its second quarter of 2018, the Company announced again revenue growth by reaching $3,228k while compared to $3,081k (5% growth) for its second quarter of 2017.
« In our second quarter of 2018, we have pursued growth in our core activities of carrier billing and communication and pursued our improvements in our various applications including VuduMobile. We have also implemented an optimization plan that would allow us to improve our financial performance in a near future. In addition, we continue to progress with regards to our strategic acquisitions and their related financing », said President and CEO of ATW Tech, Michel Guay.
The Company also announces that a total of 460 000 share purchase options have been granted at market price to officers, and consultants of the Company as well as members of the board of directors, pursuant to the terms of its share option plan (the “Plan”) and expiring on August 19, 2023.
The above data includes a summary of highlights. For further information, please consult the Corporation’s consolidated financial statement as well as the Management Report for the year ended December 31, 2017 at www.sedar.com
Forward-Looking Statements Disclaimer
This press release contains forward-looking statements that reflect the Company’s current expectation regarding future events. There is a risk that expectations and forward-looking statements will not prove to be accurate. Readers are cautioned not to place undue reliance on these forward-looking statements as they involve risks and uncertainties, which could make actual results differ materially from those projected herein and depend on a number of factors including, but not limited to, no history of profitability, future financing, intellectual property and patents, key personnel, competitive marketplace, technology obsolescence, share price volatility and other risks detailed from time to time in the Company’s filings. While ATW Tech anticipates that subsequent events and developments may cause its views to change, ATW Tech specifically disclaims any obligation to update these forward-looking statements, unless obligated to do so by applicable securities laws.
Additional information regarding the Company is available on SEDAR www.sedar.com. The TSX Venture Exchange and its Regulatory Services provider (as per meaning assigned to this term in TSX Venture Exchange’s policies) bear no liability as to the relevance or accuracy of this press release.
ABOUT ATW TECH
ATW Tech (TSX-V: ATW) is a leader in information technology, owner of several web platforms including VoxTel, VuduMobile, Atman, Bloomed and Quebec Rencontres. VoxTel offers various interactive communication and landline and mobile carrier billing phone solutions. VuduMobile is specialized the text messaging business for enterprises through its unique, user-friendly and bilingual test messaging application et turnkey solution allowing management of text message management programs in all kind of businesses. Atman and its APIs enable companies to optimize their human capital. Bloomed is a cloud-based platform to manage data (smart data) on consumers and their behaviors, which is developed for marketing agencies and their campaigns for the consumer and corporate markets. Quebec Rencontres is a web and mobile social network application catered to building serious and sustainable relationships.
| SOURCE: | |
| ATW TECH | |
| Michel Guay | Simon Bédard, CA, CPA, CFA, MBA |
| Founder, president and CEO | CFO |
| Tel.: 514.935.5959 ext. 301 | Tel. : 514.935.5959 ext. 304 |
| mguay@atwtech.com | sbedard@atmanco.com |
| www.atwtech.com |
- Published in ATWTECH
Canada Cobalt’s Re-2OX Produces 22.6% Cobalt Sulphate
Momentum Public Relations
Press Release: August 15, 2018
Canada Cobalt Works Inc. (TSXV: CCW) (OTC: CCWOF) (Frankfurt: 4T9B) (the “Company” or “Canada Cobalt”) is pleased to announce that through its proprietary Re-2OX process at SGS Lakefield, the Company has produced the first-ever premium grade cobalt sulphate from its 100%-owned Castle mine while it also moves immediately toward the creation of nickel-manganese-cobalt battery grade formulations.
Pilot plant production of cobalt-nickel-rich gravity concentrates at the Castle mine, now underway, will allow for a scaling up of the Re-2OX process this quarter.
Highlights:
- Canada Cobalt’s vertically integrated, environmentally green Re-2OX process at SGS has produced a technical grade cobalt sulphate hexahydrate at 22.6%, directly from cobalt-rich gravity concentrates produced from the first level of the Castle mine in the prolific Northern Ontario Cobalt Camp (bypassing the smelting process);
- The 22.6% grade exceeds the technical specifications of cathode producers in Asia who are in discussions with the company’s marketing representative in that region to evaluate Canada Cobalt sample product for potential battery sector use (Re-2OX will meet client specific purities);
- The very adaptable Re-2OX process will now create a Canada Cobalt suite of nickel-manganese-cobalt (NMC) battery grade formulations using an additive approach where necessary.
Frank Basa, President and CEO, commented: “Through the expertise of Dr. Ron Molnar and the team at SGS in Peterborough, CanadaCobalt has broken new ground as a technology leader in Canada’s most prolific Cobalt district. We’ve now demonstrated that from concentrate produced from the Castle mine, we can create a premium grade end product (cobalt sulphate) without a smelting process. This is a testament to the efficiency and effectiveness of Re-2OX, a process that’s very amenable to scaling up.
“We look forward to marketing the Canada Cobalt Re-2OX brand to the battery sector while we ramp up activity at the Castle mine both underground and at surface. Underground work including diamond drilling is proceeding extremely well, with another update shortly, while the pilot plant and a surface drill program to test for potential new discoveries east of the mine add important new dimensions to this exciting project,” Basa concluded.
Cobalt, nickel and manganese recoveries from the concentrate using Re-2OX were 99%, 81% and 84%, respectively, while 99% of the arsenic was removed (refer to May 31, 2018, news release).
Qualified Person
The technical information in this news release was prepared under the supervision of Frank J. Basa, P.Eng., Canada Cobalt’s President and Chief Executive Officer, who is a member of Professional Engineers Ontario and a qualified person in accordance with National Instrument 43-101.
Quality Assurance/Quality Control
An 82-kilogram sample of vein material that was taken from the first level of the Castle mine was crushed to negative 10 mesh and blended by SGS Laboratories in Lakefield, Ont., from which a representative sample was submitted for analysis by lead fusion fire assay for silver and gold. Other metals were assayed by ICP after multi-acid digestion. The material was subsequently used for testing the proprietary Re-2OX process. Canada Cobalt relies on internal SGS laboratory independent QA/QC, which allows the disclosure of the results provided.
About Canada Cobalt Works Inc.
Canada Cobalt is a pure play cobalt company focused exclusively on the Northern Ontario Cobalt Camp, Canada’s most prolific cobalt district. With three 100%-owned past producing mines, a proprietary hydrometallurgical process known as Re-2OX, and plans for a 600-tonne-per-day mill at its flagship Castle Property near Gowganda, CanadaCobalt is strategically positioned to become a vertically integrated North American leader in cobalt extraction and recovery.
“Frank J. Basa”
Frank J. Basa, P. Eng.
President and Chief Executive Officer
Neither the TSX Venture Exchange nor its Regulation Service Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release. This news release may contain forward-looking statements including but not limited to comments regarding the timing and content of upcoming work programs, geological interpretations, receipt of property titles, potential mineral recovery processes, etc. Forward-looking statements address future events and conditions and therefore, involve inherent risks and uncertainties. Actual results may differ materially from those currently anticipated in such statements.
SOURCE Canada Cobalt Works Inc.
View original content: http://www.newswire.ca/en/releases/archive/August2018/15/c6509.html
Contact:
Frank J. Basa, P.Eng., President and CEO at 1-819-797-4144
- Published in Canada Cobalt Works, Mining, News Home
Crop Advances Its San Bernardino Dispensary Application and Increases Ownership in California Farm
Momentum Public Relations
Press Release: August 15, 2018
CROP Infrastructure Corp. (CSE: CROP) (OTCMKTS: CRXPF) (“CROP” or the “Company”) announces it has received confirmation that its San Bernardino dispensary application has passed stage one of the application review process. As previously announced on July 5th 2018, CROP has agreed to finance the purchase of real estate on the granting of a California dispensary license. The proposed dispensaries will operate under the brand, “Emerald Heights”.
Furthermore, the Company is pleased to announce it has increased its ownership from 30% to 49% for its “Emerald Triangle” cannabis production facility tenanted by “Hempire” in Humboldt California. This 30,000 square foot production facility sits on approximately nine acres and consists of a 10,000 square foot medicinal cannabis greenhouse facility and an additional 20,000 square feet of recreational licensed canopy.
As previously announced on August 9th 2018, harvesting at the Humboldt operation recently began at the first of five 2,000 square foot greenhouses and is expected to continue at a rate of one greenhouse per week. As each greenhouse is harvested new starter plants will populate the canopy space. Once the 10,000 square feet of plants have been taken down, the additional 20,000 square feet of canopy will then be harvested.
CROP Infrastructure Director & CEO Michael Yorke states: “This is an exciting time for CROP as our tenant begins its first harvest in California which makes this an ideal time to increase our ownership in the project from 30% to 49%. Our Humboldt facility is located in one of the most important cannabis jurisdictions in the US and potentially the world known as the Emerald Triangle. With the advancement of our dispensary application we are now moving towards assisting tenants in becoming fully vertically integrated in the State of California.”
The Company will purchase the additional 20% of Humboldt for total consideration of $1.0 Million CAD by issuing 5,000,000 shares at a deemed price of $0.20 per share; 100% of these shares will be escrowed for 12 months, with 25% becoming free trading every 3 months thereafter.
About Humboldt Holdings LLC
Located in Humboldt County, California the property is 8.46 acres and currently houses a 10,000 square foot greenhouse as well as a barn, garage and residence. On site are five 5,000-gallon water tanks, a well and pump house and a 30 x 60 ft. drying shed. The property is zoned for a 10,000 square foot medical and a 20,000 square foot recreational cannabis license.
https://cropcorp.com/property/california/
About CROP
CROP Infrastructure Corp. is publicly listed on the Canadian Securities Exchange and trades under the symbol “CROP” and in the US under the symbol “CRXPF”. CROP is primarily engaged in the business of investing, constructing, owning and leasing greenhouse projects as part of the provision of turnkey real estate solutions for lease-to-licensed cannabis producers and processors offering best-in-class operations. The Company’s portfolio of projects includes cultivation properties in California, Washington State, Nevada, Italy, Jamaica and a joint venture on West Hollywood and San Bernardino dispensary applications. CROP has developed a portfolio of 15 Cannabis brands and has US and Italian distribution rights to a line of over 55 topical cannabis products from The Yield Growth Corp.
Company Contact
Michael Yorke – CEO & Director
E-mail: info@cropcorp.com
Website: www.cropcorp.com
Phone: (604) 484-4206
- Published in Business, CROP Infrastructure, Medical Marijuana, News Home
Genacol Canada Corporation and Tetra Bio-Pharma Team Up to bring Innovative Cannabinoid Treatments to Self-Care Market in Canada
Momentum Public Relations
Press Release: August 14 2018
Tetra Bio-Pharma Inc., a leader in cannabinoid-based drug discovery and development (TSX VENTURE: TBP) (OTCQB: TBPMF), is pleased to announce that they have concluded a Development and Commercialization agreement with Genacol Canada Corporation. Tetra Bio-Pharma will develop a cannabinoid derived oral capsule and a topical cream for treating joint pain and inflammation. Genacol will be responsible for the promotion and sales of these products throughout their network in Canada. According to industry statistics the market for topical pain relief products exceeded $2.5 billion. The companies plan to launch the products in 2019.
|
||||||||||
Tetra Bio-Pharma will use its formulation and regulatory expertise and clinical trial data from its topical cannabinoid and encapsulated cannabis oil (PPP005) trials to create innovative products for Genacol. Tetra Bio-Pharma will work with regulatory to bring these products to markets within Genacol’s sales network. “We are thrilled to partner with Genacol, a market leader in Canada with a sales network in over 40 countries, these products will command a great deal of exposure,” says Dr. Guy Chamberland, interim CEO and Chief Scientific Officer of Tetra Bio-Pharma. “Tetra is in the business of developing cannabinoid derived prescription and natural health products and this agreement will enable Tetra Bio-Pharma to expand the number of offerings to our patients.”
“We are extremely excited about the potential of this partnership we have created with Tetra Bio-Pharma, a world leader in developing cannabinoid derived prescription and natural health products,” stated Martin Vidal, President & Chief Executive Office for Genacol Corporation. “Our research team at Genacol are great believers in the therapeutic properties of cannabinoids. These products that Tetra Bio-Pharma is developing and Genacol will promote and commercialize are expected to play an important role in the pain and inflammation market in Canada. We estimate that these products will generate several millions of dollars in sales for Genacol in Canada.”
About Tetra Bio-Pharma
Tetra Bio-Pharma (TSX-V: TBP) (OTCQB: TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and development with a Health Canada approved, and FDA reviewed, clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. The Company has several subsidiaries engaged in the development of an advanced and growing pipeline of Bio Pharmaceuticals, Natural Health and Veterinary Products containing cannabis and other medicinal plant-based elements. With patients at the core of what we do, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies.
For more information visit: www.tetrabiopharma.com
About Genacol Corporation
Genacol Corporation Canada is a leader in the development and marketing of natural health products primarily related to the treatment of joint pain and the maintenance of joint health. Its mission is to improve the quality of life of its customers by offering them a range of innovative natural products. Building on its success in Canada, Genacol has also developed in recent years an international presence with Genacol brand products available in more than 40 countries, including Latin America, Europe, Asia, the Middle East, Africa and the United States. In addition, the Genacol trademark is registered in 81 countries worldwide. The success of Genacol is based on its collagen manufactured using a proprietary process called “AminoLock® Sequential Technology”, which is an ingredient in many of Genacol’s products.
For more information visit: www.genacol.ca/en/about-us/
Source: Tetra Bio-Pharma
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-looking statements
Some statements in this release may contain forward-looking information. All statements, other than of historical fact, that address activities, events or developments that the Company believes, expects or anticipates will or may occur in the future (including, without limitation, statements regarding potential acquisitions and financings) are forward-looking statements. Forward-looking statements are generally identifiable by use of the words “may”, “will”, “should”, “continue”, “expect”, “anticipate”, “estimate”, “believe”, “intend”, “plan” or “project” or the negative of these words or other variations on these words or comparable terminology. Forward-looking statements are subject to a number of risks and uncertainties, many of which are beyond the Company’s ability to control or predict, that may cause the actual results of the Company to differ materially from those discussed in the forward-looking statements. Factors that could cause actual results or events to differ materially from current expectations include, among other things, without limitation, the inability of the Company to obtain sufficient financing to execute the Company’s business plan; competition; regulation and anticipated and unanticipated costs and delays, the success of the Company’s research and development strategies, the applicability of the discoveries made therein, the successful and timely completion and uncertainties related to the regulatory process, the timing of clinical trials, the timing and outcomes of regulatory or intellectual property decisions and other risks disclosed in the Company’s public disclosure record on file with the relevant securities regulatory authorities. Although the Company has attempted to identify important factors that could cause actual results or events to differ materially from those described in forward-looking statements, there may be other factors that cause results or events not to be as anticipated, estimated or intended. Readers should not place undue reliance on forward-looking statements. While no definitive documentation has yet been signed by the parties and there is no certainty that such documentation will be signed. The forward-looking statements included in this news release are made as of the date of this news release and the Company does not undertake an obligation to publicly update such forward-looking statements to reflect new information, subsequent events or otherwise unless required by applicable securities legislation.
For further information, please contact Tetra Bio-Pharma Inc.
Robert (Bob) Bechard
Executive Vice President, Corporate Development and Licensing
514-817-2514
Investors@tetrabiopharma.com
Media Contact:
ACJ Communication
Daniel Granger
Danel.granger@acjcommunication.com
514-840-7990
514-232-1556
Photos accompanying this announcement are available at
http://www.globenewswire.com/NewsRoom/AttachmentNg/b867bc64-b347-4043-80ff-c7d022ab7eff
http://www.globenewswire.com/NewsRoom/AttachmentNg/3464aadf-745d-4d04-b230-b71f0c82188a
- Published in Tetra Bio Pharma