DIAGNOS Announces an Extension to the Diabetic Retinopathy Screening Services Assisted by Artificial Intelligence (AI) at the CHUM
Momentum Public Relations
Press Release: January 22, 2019
DIAGNOS Inc. (“DIAGNOS” or the “Company”) (TSX Venture Exchange: ADK) (OTCQB: DGNOF), a leader in early detection of critical health issues through the use of its FLAIRE platform based on Artificial Intelligence (AI), announces today, further to our initial news release dated June 5th, 2018, the extension through May 2019 of the technological showcase for the automated detection of Diabetic Retinopathy at the Centre Hospitalier de l’Université de Montréal (CHUM).
Technological showcase
DIAGNOS will continue screening for diabetic retinopathy at the CHUM until the end of May 2019. Because of excellent initial screening results, the administration of the CHUM has elected to extend the duration of the technological showcase to further appreciate the benefits.
AI for detection and triage
Our early detection service includes an automated AI analysis of fundus imagery together with a triage system according to the degree of severity. This proprietary service is only available via our CARA telehealth platform (Computer Assisted Retina Analysis).
Improving patient services
The DIAGNOS automated screening solution significantly increases accessibility for diabetic and hypertensive patients subject to annual screening. Screening is done at the service centre using fundus photography. The process is fast, painless, and very simple for the patient. The CARA platform will facilitate identification and expedited triage of patients suffering from retinopathies who have been referred to the ophthalmology department following a consultation at the endocrinology or diabetes clinic.
DIAGNOS is also announcing the resignation of Mr. Jean-Yves Thérien from his position as director and chairman of the audit committee of the Corporation due to personal reasons. “On behalf of my fellow board members, I would like to sincerely thank Jean-Yves for his dedication and important contribution to the development of DIAGNOS”, said Mr. Georges Hébert, Chairman of the Board of directors. The Corporation is in the process of finding Mr. Thérien’s replacement as chairman of the audit committee. In the interim, Mr. Hébert will assume the role of chairman of the audit committee.
About DIAGNOS
DIAGNOS has screened to date more than 250,000 diabetic patients. Thanks to the support of the Government of Quebec and our collaboration with the CHUM, this technological showcase will allow Diagnos to demonstrate our expertise in the province. We strongly believe this partnership will also help set up extensive screening campaigns in other Canadian provinces and in the United States.
About CARA
CARA is a proprietary tele-ophthalmology platform that integrates with existing equipment (hardware and software) and processes at the point of care. CARA’s Artificial Intelligence image enhancement algorithms make standard retinal images sharper, clearer, and easier to read. CARA is accessible securely over the internet, and is compatible with all recognized image formats and brands of fundus cameras, and is EMR compatible. CARA is a cost-effective tool for screening large numbers of patients in real-time and has been cleared for commercialization by several regulatory authorities such as Health Canada, the U.S. Food and Drug Administration and the European Union.
Additional information is available at www.diagnos.com and www.sedar.com
- Published in Diagnos, Life Sciences, News Home, Technology
Diagnos 900,000 shares for debt
Momentum Public Relations
Press Release: December 12, 2018
The TSX Venture Exchange has accepted for filing the company’s proposal to issue 900,000 common shares at a price of five cents per share in settlement of a total amount of $45,000 on outstanding interest payable on convertibles notes issued in October, 2017.
Number of creditors: seven creditors
Insiders: Gestion Maurice Pinsonnault (Maurice Pinsonnault), 200,000; Danie Clerk, 200,000
For further information, please refer to the company’s press releases dated Oct. 24, 2018, and Nov. 21, 2018.
- Published in Diagnos, Life Sciences, News Home, Technology
DIAGNOS is announcing a collaboration with ÉTS on Automated detection and grading of Diabetic Retinopathy
Momentum Public Relations
Press Release: December 04, 2018
Diagnos Inc. (“DIAGNOS” or “the Corporation”) (TSX Venture:ADK), (OTCQB: DGNOF), a leader in early detection of critical health issues through the use of Artificial Intelligence (AI), announces today a collaboration with ÉTS, École de technologie supérieure, an engineering school part of Université du Québec. The collaboration is funded in part by the Natural Science and Engineering Research Council of Canada (NSERC). Our focus will be to measure the benefit of using deep convolutional neural networks in detecting anomalies of diabetic retinopathy and classifying the patient by level of severity.
The main outcome of this collaboration is to bring Diagnos’ algorithms to a newlevel of automation, speed and accuracy. “Diagnos is always interested in cutting-edge algorithms in deep learning and with this collaboration, this will enable Diagnos to engage with leading academics to develop state-of-the-art screening solutions,” said André Larente, CEO at Diagnos Inc.
Under the supervision of Professor Ismail Ben Ayed, ÉTS students will work with Diagnos in using deep convolutional neural networks to detect anomalies caused by diabetic retinopathy and classify the patients by level of severity. The images will be provided by our CARA (Computer Assisted Retina Analysis) database. “A data-driven, large-scale and optimization based approach, which promises to deliver retinal image interpretation algorithms with a much higher practical impact,” said Professor Ismail Ben Ayed.
We anticipate that this partnership will increase automation, efficiency and precision for early identification and monitoring of diabetic retinopathy. “Identifying patients in early stages of diabetic retinopathy is our main goal because we can treat them efficiently. Our objective is to refer patients at high risk of going blind,” said Dr Hadi Chakor, CMO at Diagnos Inc.
This is our second collaboration project in Quebec this year. “We are deeply committed to develop our ties in our community. Our technology project with CHUM (Centre Hospitalier Universitaire de Montréal) was our first large investment in a teaching hospital. This announcement, with the financial support of Natural Science and Engineering Research Council of Canada (NSERC), represents the second step of our strategy to collaborate with an engineering school to foster innovation and speed up our go to market plan. We are building the case from which to grab market share in the North America healthcare market,” said Yves-Stephane Couture, Vice-President of Sales at Diagnos.
About ETS
A University that Specializes in Applied Engineering. The École de technologie supérieure is a constituent establishment of the Université du Québec. ÉTS, which specializes in engineering and technological transfer education as well as applied research, trains engineers and researchers who are recognized for their practical and innovative approach. Since its creation, ÉTS has pursued a mission that is deeply rooted in all its activities: To meet the needs of the industrial sector, which is in need of engineers who have not only a good theoretical background, but also practical knowledge. To fulfil this mission, ÉTS has a unique partnership with the business and industrial spheres that includes both small and large companies. It stands out from other universities in Québec because of the applied training it offers students, as well as its research activities conducted by and for companies.
About DIAGNOS
DIAGNOS is a publicly-traded Canadian corporation with a mission of early detection of critical health issues through the use of its Artificial Intelligence (“AI”) tool CARA (Computer Assisted Retina Analysis). CARA is a tele-ophthalmology platform that integrates with existing equipment (hardware and software) and processes at the point of care. CARA’s Artificial Intelligence image enhancement algorithms make standard retinal images sharper, clearer and easier to read. CARA is accessible securely over the internet, and is compatible with all recognized image formats and brands of fundus cameras, and is EMR compatible. CARA is a cost-effective tool for screening large numbers of patients in real-time and has been cleared for commercialization by several regulatory authorities such as Health Canada, the U.S. Food and Drug Administration and the European Union.
Additional information is available at www.diagnos.com and www.sedar.com
For further information, please contact:
| Mr. André Larente, President | Josh Falle |
| DIAGNOS Inc. | Momentum PR |
| Tel: 450-678-8882 ext. 224 | Tel: 514-416-4656 |
| alarente@diagnos.ca | josh@momentumpr.com |
- Published in Business, Diagnos, Life Sciences, News Home, Technology
MedX Health completes $182,000 closing of placement
Momentum Public Relations
Press Release: November 23, 2018
MedX Health Corp. (“MedX” or the “Company”) (TSX-V:MDX) announces that, pursuant to a non-brokered Private Placement Offering (the “Placement Offering”), it has completed an additional Closing of 1,137,500 Units on November 22, 2018, to raise $182,000 from accredited investors pursuant to a Private Placement Offering originally announced on October 11, 2018. Each Unit, priced at $0.16, comprises one fully paid common share and one share purchase warrant; each warrant entitles the holder to purchase one additional share at a price of $0.35 during the period up to three years from the closing date of the placement. The securities issued as a result of the Placement Offering will be restricted from trading for four months following each Closing.
Following this Closing, MedX has 135,053,901 common shares issued and outstanding.
The Company also announces that it is initiating a new, non-brokered, Private Placement offering of Units to raise up to $2.5 million from accredited investors. Each Unit in this proposed new placement will be priced at $0.16, and will comprise one fully paid common share and one share purchase warrant; each warrant will entitle the holder to purchase one additional share at a price of $0.35 during the period up to three years from the closing date of the placement. Closing of the proposed new Placement Offering is subject to all relevant regulatory and other consents and approvals, including acceptance by the TSX Venture Exchange.
About MedX
MedX, headquartered in Mississauga, Ontario, is a leading medical device and software company focused on skin cancer with its DermSecure™ telemedicine platform, utilizing its SIAscopy technology. SIAscopy is also imbedded in its products SIAMETRICS™, SIMSYS™, and MoleMate™, which MedX manufactures in its ISO 13485 certified facility. SIAMETRICS™, SIMSYS™, and MoleMate™ include hand-held devices that use patented technology utilizing light and its remittance to view up to 2 mm beneath suspicious moles and lesions in a pain free, non-invasive manner, with its software then creating real-time images for physicians and dermatologists to evaluate all types of moles or lesions within seconds. These products are Health Canada, FDA (US), ARTG and CE cleared for use in Canada, the US, Australia, New Zealand, the European Union and Turkey. MedX also designs, manufactures and distributes quality photobiomodulation therapeutic and dental lasers to provide drug-free and non-invasive treatment of tissue damage and pain. www.medxhealth.com.
This press release does not constitute an offer of any securities for sale. This press release contains certain forward-looking statements within the meaning of applicable Canadian securities legislation. These forward-looking statements involve certain risks and uncertainties that could cause actual results to differ, including, without limitation, the company’s limited operating history and history of losses, the inability to successfully obtain further funding, the inability to raise capital on terms acceptable to the company, the inability to compete effectively in the marketplace, the inability to complete the proposed acquisition and such other risks that could cause the actual results to differ materially from those contained in the company’s projections or forward-looking statements. All forward looking statements in this press release are based on information available to the company as of the date hereof, and the company undertakes no obligation to update forward-looking statements to reflect events or circumstances occurring after the date of this press release.
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this press release.
View source version on businesswire.com: https://www.businesswire.com/news/home/20181123005259/en/
Contacts:
Scott Spearn, President and CEO
MedX Health Corp
905-670-4428 ext 229
Media Relations
Deborah Thompson
dthompson@medxhealth.com
416-918-9551
- Published in Life Sciences, MedX Health Corp., News Home, Technology
DIAGNOS Announces Final Closing of Private Placement
Momentum Public Relations
Press Release: November 21, 2018
DIAGNOS Inc. (“DIAGNOS”, the “Corporation” or the “Issuer”) (TSX Venture: ADK) (OTCQB: DGNOF), a leader in early detection of critical health issues through the use of Artificial Intelligence, announces the final closing of a private placement (“Private Placement”) of 20,660,000 units (each a “Unit”) issued at $0.05 per Unit for gross proceeds of $1,033,000 following receipt of required regulatory approvals. The Private Placement was announced on November 6, 2018 and on November 9, 2018.
All monies quoted in this press release shall be stated and paid in lawful money of Canada.
About DIAGNOS
DIAGNOS is a publicly-traded Canadian corporation with a mission of early detection of critical health issues through the use of its Artificial Intelligence (“AI”) tool CARA (Computer Assisted Retina Analysis). CARA is a tele-ophthalmology platform that integrates with existing equipment (hardware and software) and processes at the point of care. CARA’s Artificial Intelligence image enhancement algorithms make standard retinal images sharper, clearer and easier to read. CARA is accessible securely over the internet, and is compatible with all recognized image formats and brands of fundus cameras, and is EMR compatible. CARA is a cost-effective tool for screening large numbers of patients in real-time and has been cleared for commercialization by several regulatory authorities such as Health Canada, the U.S. Food and Drug Administration and the European Union.
Additional information is available at www.diagnos.com and www.sedar.com.
For further information, please contact:
| Mr. André Larente, President | Josh Falle | ||
| DIAGNOS Inc. | Momentum PR | ||
| Tel: 450-678-8882 ext. 224 | Tel: 514-416-4656 | ||
| alarente@diagnos.ca | josh@momentumpr.com | ||
- Published in Diagnos, Life Sciences, News Home, Technology
DIAGNOS Enters into a Shares for Debt Transaction
Momentum Public Relations
Press Release: November 21, 2018
DIAGNOS Inc. (“DIAGNOS” or the “Corporation”) (TSX Venture: ADK) (OTCQB: DGNOF), a leader in early detection of critical health issues through the use of Artificial Intelligence, announces today it has entered into a shares for debt arrangement in which the Corporation is issuing 900,000 common shares (each a “Share”), at a deemed price of $0.05 per Share, in settlement for interests payable in the amount of $45,000 on outstanding unsecured convertible notes (each a “Note”) totaling $900,000.
Satisfying this outstanding indebtedness with shares was undertaken in order to preserve the Corporation’s cash for operational purposes.
Two Note holders eligible to receive common shares as part of the shares for debt transaction are considered “related parties” of the Corporation within the meaning of Multilateral Instrument 61-101 – Protection of Minority Security Holders in Special Transactions (“MI 61-101”). One person related to one Note holder, as well as one other Note holder will each receive 200,000 common shares as part of the transaction. The transaction is exempt from the valuation requirement and the minority approval requirement prescribed in MI 61-101 based on the fact that the fair market value of the related parties’ participations in the shares for debt transaction does not exceed 25% of DIAGNOS’ market capitalization.
Shares issued as part of the shares for debt arrangement are subject to a statutory four-month hold period from the date of issuance ending March 21, 2019.
The issuance of shares is subject to receipt of all required approvals, including the approval of the TSX Venture Exchange, as well as the execution of formal documentation.
All monies quoted in this press release shall be stated and paid in lawful money of Canada.
About DIAGNOS
DIAGNOS is a publicly-traded Canadian corporation with a mission of early detection of critical health issues through the use of its Artificial Intelligence (“AI”) tool CARA (Computer Assisted Retina Analysis). CARA is a tele-ophthalmology platform that integrates with existing equipment (hardware and software) and processes at the point of care. CARA’s Artificial Intelligence image enhancement algorithms make standard retinal images sharper, clearer and easier to read. CARA is accessible securely over the internet, and is compatible with all recognized image formats and brands of fundus cameras, and is EMR compatible. CARA is a cost-effective tool for screening large numbers of patients in real-time and has been cleared for commercialization by several regulatory authorities such as Health Canada, the U.S. Food and Drug Administration and the European Union.
Additional information is available at www.diagnos.com and www.sedar.com.
For further information, please contact:
| Mr. André Larente, President | Josh Falle |
| DIAGNOS Inc. | Momentum PR |
| Tel: 450-678-8882 ext. 224 | Tel: 514-416-4656 |
| alarente@diagnos.ca | josh@momentumpr.com |
- Published in Diagnos, Life Sciences, News Home, Technology
ATW Tech Provides Additional Information Regarding Signature of a Letter of Intent for Major Acquisition of Greywolf Entertainment Group
Momentum Public Relations
Press Release: November 16, 2018
ATW Tech Inc. (“ATW Tech”) (TSX-V: ATW) has, as previously announced on November 6, 2018, signed a letter of intent for the acquisition from Fastrack Trust (the “Trust”) of all of the outstanding shares of 6855318 Canada Ltd. and 6890202 Canada Ltd. (“Greywolf Entertainment Group” or “Greywolf”). For the unaudited financial year ended December 31, 2017, Greywolf’s revenues were $10.0 million ($11.8 million in 2016) for a net income of $196k ($350k in 2016), with total assets of $2.5 million ($2.4 million) and negative shareholder’ equity of $914k ($759k).
Greywolf Entertainment Group, based in Calgary, provides turnkey full commercial production for interactive TV and/or voting networks required for such shows for landline and mobile consumers. Greywolf’s subsidiaries include Triton Global Business Services Inc. (“Triton”) and Fastrack Global Billing Networks Inc. (“Fastrack”). Triton is a North American clearinghouse that provide billing and networks for phone bills in North America and over 40 other countries. Triton has been listed in the top 300 technology companies from Canada several times. Fastrack is a North American inter-exchange carrier providing networks to its switch centers that intelligently routes consumer traffic to optimized billing or response solutions. Its network is designed to handle mass calling for both short and long periods of time, using its own robust backbone Standard Internet Protocol (SIP) networks to transport traffic anywhere in the world.
The letter of intent contemplates a purchase price of $2.95 million, which is subject to certain adjustments and payable through a cash payment of $2.0 million at closing, a vendor take-back note of $0.3 million payable in 30 equal monthly installments and $0.65 million worth of common shares of ATW Tech. No commission is payable and no change of control will result.
“We are proud to come to an agreement on this strategic acquisition of Greywolf that will help us consolidate our position in the Canadian carrier billing market, accelerate our growth, while almost doubling our revenues, and benefiting from significant cost synergies. Along with the divestiture of the Atman division completed on October 31, 2018, which represented 9.5% of ATW Tech’s revenues in 2017, this contemplated acquisition reflects our will and strategy to concentrate our activities in the financial technologies (“fintech”), particularly on our core integrated payment, interactive communication and voting platforms. This acquisition shall be the first step of our acquisition growth plan with an international reach expected for 2019” said Michel Guay, President and CEO of ATW Tech.
The closing of this transaction between ATW Tech and Greywolf, which are dealing at arm’s length, is conditional among other things on ATW Tech carrying out a satisfactory due diligence on Greywolf, obtaining satisfactory financing and all necessary regulatory approvals (including that of the TSX Venture Exchange). Closing of this transaction is expected before the end of 2018.
The Company also announces that a total of 900 000 share purchase options have been granted to a consultant of the Company at a strike price of $0.10 pursuant to the terms of its share option plan (the “Plan”) and expiring on November 15, 2023.
- Published in Atmanco, ATWTECH, Business, Financial Technology, News Home, Technology
AREV Brands International Ltd. announces Success in Terpene Blends on 1st Degree Burns and Insect Bites
Momentum Public Relations
Press Release: November 14, 2018
AREV Brands International Ltd. (” AREV ” or the “Company”) ( CSE – AREV ), is pleased to announce AREV Brands International Ltd. has has reached another major milestone in one of its several joint development projects with Alternative Extracts Inc. Further to the research work reported on September 20, 2018, terpenes from non-regulated sources were used to formulate a proprietary composition for burns and insect bites. As noted in peer-reviewed literature, terpene synthesis is vital for cannabinoid synthesis as it provides the GPP molecule for CBGA synthesis.
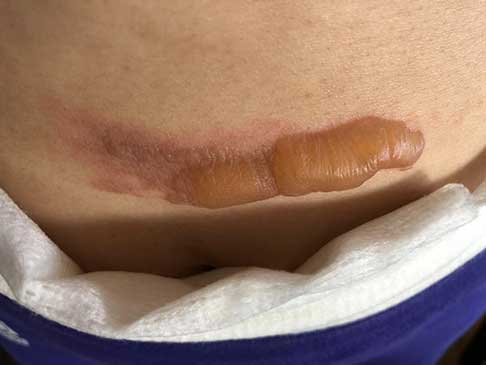
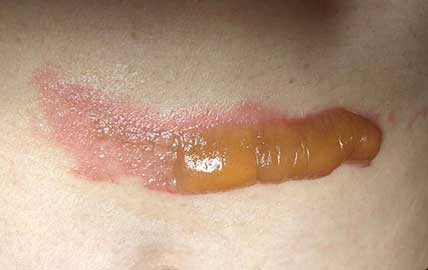
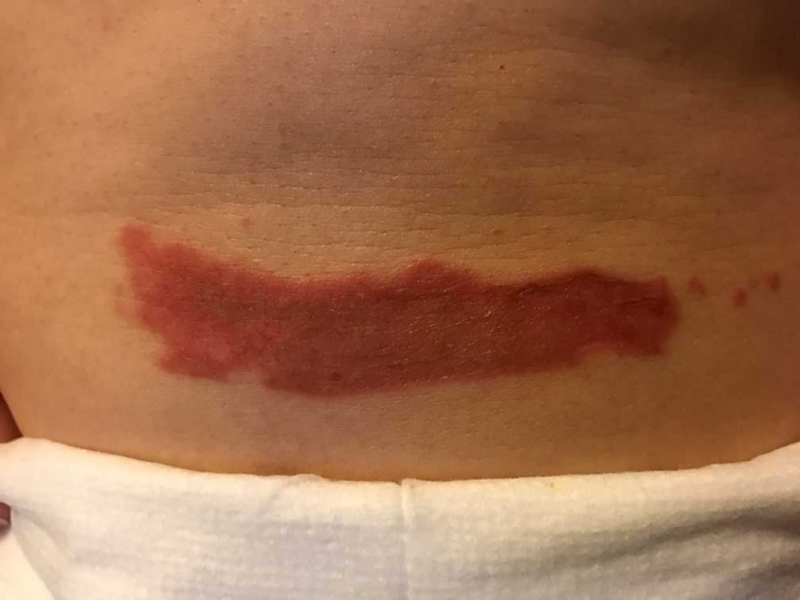
As evidenced in these images (which was first taken within 18 hours of the burn accident, and in 24-hour succession thereafter), the blisters from the burn were completely eliminated within 4 days from the first application. The patient in this case is a 50-year old woman who has a known history of keloid formation. It is noted that the patient did not develop any infection.
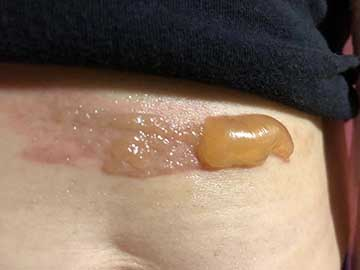
AREV extracted the valuable terpenes from selected non-regulated plant materials and exudates from trees using its extraction systems that it acquired from Alternative Extracts Inc. The composition was formulated in collaboration between AREV and AEI. The mechanism of the composition is believed to be the result of the poly-molecular function of the terpenes.
The company cautions, that although these results are encouraging, a wider sample population is required. A formal study will be conducted in the coming months.
Burn data from World Health Organization shows that:
- – In India, over 1,000,000 people are moderately or severely burnt every year.
– Nearly 173,000 Bangladeshi children are moderately or severely burnt every year.
– In Bangladesh, Colombia, Egypt and Pakistan, 17% of children with burns have a temporary disability and 18% have a permanent disability.
– Burns are the second most common injury in rural Nepal, accounting for 5% of disabilities.
– In 2008, over 410,000 burn injuries occurred in the United States of America, with approximately 40,000 requiring hospitalization.
Mike Withrow commented “Although we are pleased to have been able to minimize the pain and scarring in this burn patient, the results from the use of our proprietary blend of natural compounds warrants further testing and trials.”
Dr. Adrian Wade, Chief Science Officer, of Alternative Extracts Inc. commented, “This collaboration continues to produce highly effective compounds and formulations. Terpenes have demonstrated to play an important role in an increasing number of natural health and medical products.”
Seek safe harbor and forward looking statements.
For further information, contact Mike Withrow, mike.withrow.vn@gmail.com, (778) 896-6536?.
On behalf of the Board,
Mike Withrow
CEO & Director
About AREV Brands International Ltd.
AREV Brands International Ltd. (“AREV”) produces and delivers functional compounds and ingredients from its world-class extraction systems. AREV is revolutionizing the current delivery method of terpenes, cannabinoids and flavonoids. These premium ingredients and formulations are used in products targeted for sale in the natural health, medical, functional food, nutraceutical, sport nutrition and bioceutical markets. AREV innovates through extraction to produce extracts from specific selected plant and exude from trees that address 5 areas of health including Anxiety, Pain Management, Insomnia, Central Nervous System Disorders & Libido.
- Published in AREV Nutrition Sciences, Business, Life Sciences, Medical Marijuana, News Home, Technology
DIAGNOS Announces Today the Addition of a Strategic Partner in the Country of Mexico
Momentum Public Relations
Press Release: November 13, 2018
DIAGNOS Inc. (“DIAGNOS” or “the Corporation”) (TSX Venture:ADK), (OTCQB: DGNOF), a leader in early detection of critical health issues through the use of Artificial Intelligence (AI), announces today the addition of a strategic partner in the country of Mexico. This agreement will enlarge the footprint coverage of DIAGNOS’ offering for monitoring diabetic patients through our first partner in that country.
“Today we all (my family and I), accepted unanimously to go with DIAGNOS in this adventure, we truly believe both of us will do great and together will achieve important objectives”, declares Quim. Sergio Galindo, founder and owner of Axmilab. “Mr. Andre Larente visited me in Monterrey city, my hometown at my brand new office, we clicked right away because we both are men of one vision: to provide the best diagnostic technology to prevent and leverage the health quality of people.” “I also met Mr. Georges Herbert Chairman of DIAGNOS Inc, during his visit to Mexico City. Very important reasons for me to partner with DIAGNOS, because I partner with true people”, finished Quim. Sergio Galindo.
“We express our gratitude and celebrate along with Axmilab family their decision to collaborate with us, the first step is to prepare ourselves for key projects in different states of Mexico”, mentions Mr. Guillermo Moreno, Vice President of DIAGNOS, Latin America.
About AXIMILAB
Axmilab is a Mexican company with more than 24 years of experience by manufacturing and distributing products in the health sector. His founder, Quim. Sergio Galindo, has successfully driven the business since its conception in 1994, now with the support of his family, is growing exponentially as an example, they’ve been a supplier for IMSS (by winning contracts in the last 13 consecutive years in the diabetes area). Its dominant territory is Northern Mexico, headquarters is in Monterrey, N.L., also covering Mexico City, Sonora, Queretaro, Chiapas, Chihuahua, Durango, Jalisco, Yucatan, San Luis Potosi States among others. Since 1998, Axmilab has been working at all levels of Government, also represents major brands such as Roche, J&J, Licon, PKL, Alifax.
About DIAGNOS
DIAGNOS is a publicly-traded Canadian corporation with a mission of early detection of critical health issues through the use of its Artificial Intelligence (“AI”) tool CARA (Computer Assisted Retina Analysis). CARA is a tele-ophthalmology platform that integrates with existing equipment (hardware and software) and processes at the point of care. CARA’s Artificial Intelligence image enhancement algorithms make standard retinal images sharper, clearer and easier to read. CARA is accessible securely over the internet, and is compatible with all recognized image formats and brands of fundus cameras, and is EMR compatible. CARA is a cost-effective tool for screening large numbers of patients in real-time and has been cleared for commercialization by several regulatory authorities such as Health Canada, the U.S. Food and Drug Administration and the European Union.
Additional information is available at www.diagnos.com and www.sedar.com
For further information, please contact:
| Mr. André Larente, President | Josh Falle |
| DIAGNOS Inc. | Momentum PR |
| Tel: 450-678-8882 ext. 224 | Tel: 514-416-4656 |
| alarente@diagnos.ca | josh@momentumpr.com |
- Published in Diagnos, Life Sciences, News Home, Technology
AREV Brands International Ltd. Announces Asset Purchase Agreement Completed to Bare Topicals from Alternative Extracts Inc.
Momentum Public Relations
Press Release: November 9, 2018
AREV Brands International Ltd. (” AREV ” or the “Company”) ( CSE – AREV ), is pleased to announce it has completed the asset purchase (the “Acquisition Agreement”) dated November 9, 2018 with Alternative Extracts Inc. (“AEI”). To acquire 100% of the Bare Topicals assets in consideration for issuance of 500,000 common shares of the Company at a deemed price of $0.32 per share issued by the Company, for an aggregate value of $160,000.
Bare Brands is a Line of Award Winning Cannabis Infused Topical Products aim to provides safe alternatives to achieving significant pain relief and treatment of skin conditions.
2016
Bio Cup – 1st place topical (Magik)
Bio Cup – 2nd Place topical (SPF 420)
Bio Cup – 1st place topical people’s choice (Magik)
Bio Cup – 2nd Place topical people’s choice (SPF 420)
Bio Cup – 2nd Place best product (overall for the event) (SPF 420)
Karma Cup – 3rd place topical (Magik)
2017
Okanagan Cannabis Cup – 2nd place topical (Magik)
High Times Cannabis Cup – 2nd Place topical (Magik)
Karma Cup – 1st place topical (Recover)
Karma Cup – 3rd place topical (Magik)
2018
The Canna Cup – 1st place topical
The Grey Area Festival – 2nd Place topical (Releaf)
Okanagan Cannabis Cup – 2nd Place topical (Recover)
Karma Cup – 2nd Place topical (Recover)
The most awarded topical company in Canada.
Mike Withrow, CEO for AREV Brands International states “This acquisition is another that adds to the portfolio of products AREV continues to assemble while working on obtaining ACMPR Licencing to sell any of our cannabis based products. In the meantime we are preparing the Bare Topicals herbal line for production.”
The Company, effective immediately, has issued 500,000 stock options to Directors, Officers and Consultants of the Company at a price of $0.41 for a period of 5 years from the issuance date. These options will vest immediately.
Seek safe harbor and forward-looking statements.
For further information, contact Mike Withrow, mike.withrow.vn@gmail.com.
On behalf of the Board,
Mike Withrow
CEO & Director
About AREV Brands International Ltd.
AREV Brands International Ltd. (“AREV”) produces and delivers functional compounds and ingredients from its world-class extraction systems. AREV is revolutionizing the current delivery method of terpenes, cannabinoids and flavonoids. These premium ingredients and formulations are used in products targeted for sale in the natural health, medical, functional food, nutraceutical, sport nutrition and bioceutical markets. AREV innovates through extraction to produce extracts from specific selected plant and exude from trees that address 5 areas of health including Anxiety, Pain Management, Insomnia, Central Nervous System Disorders & Libido.
- Published in AREV Nutrition Sciences, Business, Life Sciences, Medical Marijuana, News Home, Technology