Cannara Biotech – www.shopCBD.com
Cannara Biotech (CSE:LOVE)(FRA:8CB)
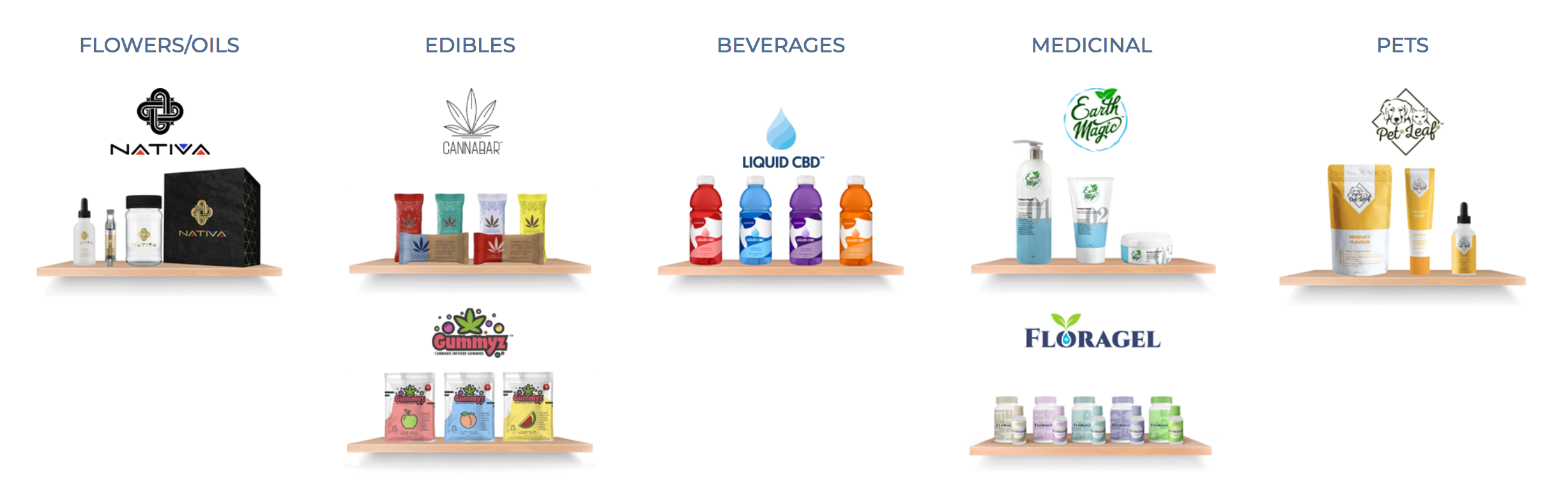
Headquartered in Montreal, Cannara Biotech is entering the U.S. THC-CBD market, through a subsidiary, with an on-line e-commerce platform called shopCBD.
“We are taking a leadership role by creating a hub for vendors to showcase their products and consumers to purchase hemp-CBD products in a fast and easy way. We want to become the Amazon of CBD,” – ZOHAR KRIVOROT – CEO and Founder
Highlights
- Zohar Krivorot, president and chief executive officer of Cannara Biotech is a 15-year veteran in the tech and on-line industries.
- Cannara Biotech is building the largest indoor cannabis cultivation facility in Quebec and one of the largest in Canada
- shopcbd.com is a platform that will showcase hemp-CBD retail products including tinctures, oils, capsules, body care, vape cartridges and pet-related CBD infused treats
- The indoor facility offers lower risk of airborne infections, mold/mildew and produces a higher grade of cannabis
- The new e-commerce platform will showcase retail products from hemp-CBD manufacturers for the U.S. consumer market
- shopCBD will provide a user-friendly on-line experience where consumers can purchase, review and compare a variety of CBD products
Trending
- With the recent passing of the 2018 Farm Bill in the U.S., CBD represents an emerging sector with many vendors seeking national reach to U.S. customers.
- The company’s strategy is to offer a wide variety of products from leading CBD brands, with competitive pricing and delivery times across the U.S.
- The rise in popularity for CBD products is driven by consumers seeking natural-based health and wellness solutions to treat various ailments from inflammation, anxiety to insomnia amongst many others.
- The hemp-derived CBD market is expected to reach $22-billion (U.S.) by 2022 according to industry analysts at Brightfield Group.
Alongside the e-commerce, on-line platform, Cannara Biotech will have on-site independent THC and CBD brands, following the adoption of regulations on cannabis-infused edibles and beverages, that will produce and ship from the facility. Cannara Biotech’s facility will be a multi-purpose facility that will cultivate recreational cannabis and produce cannabinoid-infused products including edibles, cosmetics, pet products and beverages. These products will be shipped to stores and retail outlets in Canada and to international markets.
Cannara Biotech went public in Q1 of this year and is being traded on the CSE under the ticker “LOVE” and on the FRA under the ticker “8CB”. As of February 12th, 2019, LOVE closed at .22.
—
Isabelle Arsenault
Media Relations
MomentumPR
- Published in Cannara, Marijuana, Medical Marijuana
Cannara Biotech – Quebec’s largest cultivation facility
Cannara Biotech (CSE:LOVE)(FRA:8CB)

Headquartered in Montreal, Cannara Biotech is building the largest indoor cannabis cultivation facility in Quebec and one of the largest in Canada, a modern and secure 625,000-square-foot facility in Farnham, Quebec.
“Being in Quebec allows us to take advantage of the low cost of electricity and gives us a cost advantage over our competitors.” – ZOHAR KRIVOROT – CEO and Founder
Highlights
- Some 100,000 kg of cannabis production is expected per year once all 3 phases are operational. Today, phase 1 is being built with completion expected end of Q2 2019. Once completed, yield is expected at 15,000 kg.
- Cannara Biotech has raised $55M to date, has acquired 19 provisional patents for product exclusivity, and has developed 8 assorted THC/CBD infused product brands to add to their offerings.
- Low electricity cost in Quebec as well as reduced labour cost compared to the rest of Canada gives the Company a cost advantage over their competitors.
- Independent THC and CBD brands will be produced on-site accelerating speed to market.
- The Company will leverage the facility with joint-ventures to produce edibles, pet-products, cosmetics and beverages once the processing license is secured which is estimated for the end of 2019.
- The advantage of growing in an indoor facility offers lower risk of airborne infections, mold/mildew and produces a higher grade of cannabis.
- Cannara Biotech is building an online e-commerce platform (shopCbd.com) that will serve as a podium and distribution channel for US-CBD brands for the American market.

Closer look
Phase 1 will provide 130,000ft2 of growing space. Growing is expected to commence in the fall of 2019 with an estimated yield of 15,000kg.
Cannara Biotech raised $55 million from private investors during the introduction of this project and still have over $40 million in the bank with Phase 1 fully funded.
Phase 2 forecasted to be running by 2020 is 205,000ft2 and likely to yield 38,000kg per year. Phase 3 aiming to be operational in 2022 is 277,000ft2 and expected to produce 55,000kg per year. Cannara Biotech is looking to produce over 100,000kg per year once fully operational.
Currently, 300 sq. ft. space for Phase 2 is being rented by occupants that contribute to a rental revenue of $2 million per year. These earnings pay the monthly fee for the whole facility.
Cannara Biotech’s strategy is also to maximize on the low electricity rates Quebec has with its Hydro and low labour rates in Quebec.
Growing indoor cannabis often results in a higher grade of cannabis. Growing in a controlled environment, protected from many outdoor ailments and mildew creates a more consistent and desirable product for buyers and consumers.
Alongside selling cannabis to intermediaries who will transform the crop into their desired product, Cannara Biotech will have on-site independent THC and CBD brands, following the adoption of regulations on cannabis-infused edibles and beverages, that will produce and ship from the facility. Cannara Biotech’s facility will be a multi-purpose facility that will cultivate recreational cannabis and produce cannabinoid-infused products including edibles, cosmetics, pet products and beverages. These products will be shipped to stores and retail outlets in Canada and to international markets.
Cannara Biotech went public in Q1 of this year and is being traded on the CSE under the ticker “LOVE” and on the FRA under the ticker “8CB”. As of February 12th, 2019, LOVE closed at .22.
—
Isabelle Arsenault
Media Relations
MomentumPR
Tetra BioPharma Signs Second Commercialization Deal with Azevedos Industria Farmaceutica, S.A. for the Lead RX product PPP001
Momentum Public Relations
Press Release: May 4 2018
Tetra Bio-Pharma Inc. (“Tetra” or the “Company”) (TSX-V:TBP) (OTCQB:TBPMF), and Azevedos Indústria Farmacêutica, S.A. announced that they have signed a binding term sheet for the marketing and distribution of PPP001 in Portugal. This binding term sheet will pave the way towards signing a Definitive Distribution Agreement.
Tetra is eligible to receive an upfront payment, milestone payments and will be paid a share of the profits generated by the sales of PPP001 in Portugal. Azevedos will also be responsible for registering the product, as well as all marketing and distribution in Portugal.
“We are honored to partner with Azevedos, a company established in 1775, rich in tradition, and a major player in the Portuguese pharmaceutical market. This Partnership deal provides Tetra with its foray into the European market.” stated Dr. Guy Chamberland M.Sc., Ph.D., Interim CEO and Chief Scientific Officer.
“These partnerships are very important for Azevedos Indústria Farmacêutica and mean that international entities recognize our history, values, work and our technology know-how. Although having a long past, Azevedos is a pharmaceutical company turned to the future and focused on new challenges”, says Thebar Miranda, CEO of Azevedos Indústria Farmaceutica, S.A.
About PPP001
On April 4, 2018, Tetra officially started the Phase 3 trial for PPP001 indicated for terminal stage cancer patients with a goal to improving the quality of life of these patients as well as minimizing their pain. PPP001 is being developed to be the first smokable cannabis product for advanced cancer pain available under prescription.
About Azevedos Indústria Farmacêutica, S.A
Azevedos Group is a two-century Portuguese pharmaceutical brand, whose wide scope of activity ranges from development to distribution, leading manufacturing and exports to more than 60 regulated countries worldwide. Azevedos owns more than 300 MAs for the most relevant therapeutic areas covering all technology forms manufactured at its state of the art plant holding the most recognized certifications.
About Tetra Bio-Pharma: Tetra Bio-Pharma (TSX-V:TBP) (OTCQB:TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and clinical development. Tetra is focusing on three core business pillars: clinical research, pharmaceutical promotion and retail commercialization of cannabinoid-based products. Tetra Bio-Pharma is currently developing a pipeline of five cannabinoid-based products using different delivery systems such as smokable pellets, oral tablets, eye drops and topical ointments.
More information at: www.tetrabiopharma.com
Source: Tetra Bio-Pharma
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-looking statements
Some statements in this release may contain forward-looking information. All statements, other than of historical fact, that address activities, events or developments that the Company believes, expects or anticipates will or may occur in the future (including, without limitation, statements regarding potential acquisitions and financings) are forward-looking statements. Forward-looking statements are generally identifiable by use of the words “may”, “will”, “should”, “continue”, “expect”, “anticipate”, “estimate”, “believe”, “intend”, “plan” or “project” or the negative of these words or other variations on these words or comparable terminology. Forward-looking statements are subject to a number of risks and uncertainties, many of which are beyond the Company’s ability to control or predict, that may cause the actual results of the Company to differ materially from those discussed in the forward-looking statements. Factors that could cause actual results or events to differ materially from current expectations include, among other things, without limitation, the inability of the Company, through its wholly-owned subsidiary, GrowPros MMP Inc., to obtain a license for the production of medical marijuana; failure to obtain sufficient financing to execute the Company’s business plan; competition; regulation and anticipated and unanticipated costs and delays, the success of the Company’s research strategies, the applicability of the discoveries made therein, the successful and timely completion and uncertainties related to the regulatory process, the timing of clinical trials, the timing and outcomes of regulatory or intellectual property decisions and other risks disclosed in the Company’s public disclosure record on file with the relevant securities regulatory authorities. Although the Company has attempted to identify important factors that could cause actual results or events to differ materially from those described in forward-looking statements, there may be other factors that cause results or events not to be as anticipated, estimated or intended. Readers should not place undue reliance on forward-looking statements. While no definitive documentation has yet been signed by the parties and there is no certainty that such documentation will be signed The forward-looking statements included in this news release are made as of the date of this news release and the Company does not undertake an obligation to publicly update such forward-looking statements to reflect new information, subsequent events or otherwise unless required by applicable securities legislation.
For further information, please contact:
Tetra Bio-Pharma Inc.
Dr Guy Chamberland, interim CEO and CSO
guy@tetrabiopharma.com
514-220-9225
Robert (Bob) Bechard, MBA, MSc., BA,
Vice-President Finance and Business Development
bob@tetrabiopharma.com
514-817-2514
For media information, please contact:
Daniel Granger
Daniel.granger@acjcommunication.com
ACJ Communication
O: 1 514-840-7990 M: 1 514-232-1556
- Published in Tetra Bio Pharma
Tetra Bio-Pharma Signs Landmark Commercialization Term Sheet for its Lead Pharmaceutical Product, PPP001, in Israel
Momentum Public Relations
Press Release: April 24
Tetra Bio-Pharma Inc. (“Tetra” or the “Company”) (TSX VENTURE:TBP) (OTCQB:TBPMF), announced today that the Company has signed a first binding term sheet for the marketing and distribution of PPP001 in Israel with Kamada Ltd., a leading pharmaceutical company. The signing of a Definitive Distribution Agreement is expected to follow shortly. PPP001 is being developed to be the first smokable cannabis product for advanced cancer pain available under prescription.
This first international market commercialization agreement represents a significant milestone and a validation of Tetra Bio-Pharma’s business model with a leading Israel-based pharmaceutical company. Israel, like Canada, is considered one of the world leaders in the production and development of cannabinoid-based products. Kamada is a company with two FDA-approved products and an Israeli-based distribution segment that has demonstrated continued growth. Tetra Bio-Pharma intends to work closely with Kamada as PPP001 advances towards regulatory approval and commercial launch in Israel.
Under the terms of the anticipated final agreement, Kamada will be responsible for registering the product, as well as all marketing and distribution, in Israel. Tetra will be eligible to receive certain milestone payments and an undisclosed percentage of the sales of PPP001 generated by Kamada in Israel.
About PPP001
On April 4, 2018, Tetra Bio-Pharma officially started the Phase 3 trial for PPP001 indicated for terminal stage cancer patients with a goal to improving the quality of life of these patients as well as minimizing their pain. PPP001 is being developed to be the first smokable cannabis product for advanced cancer pain available under prescription.
About Tetra Bio-Pharma: Tetra Bio-Pharma (TSX VENTURE:TBP) (OTCQB:TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and clinical development. Tetra is focusing on three core business pillars: clinical research, pharmaceutical promotion and retail commercialization of cannabinoid-based products. Tetra Bio-Pharma is currently developing a pipeline of five cannabinoid-based products using different delivery systems such as smokable pellets, oral tablets, eye drops and topical ointments. More information at: www.tetrabiopharma.com
Source: Tetra Bio-Pharma
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-looking statements
Some statements in this release may contain forward-looking information. All statements, other than of historical fact, that address activities, events or developments that the Company believes, expects or anticipates will or may occur in the future (including, without limitation, statements regarding potential acquisitions and financings) are forward-looking statements. Forward-looking statements are generally identifiable by use of the words “may”, “will”, “should”, “continue”, “expect”, “anticipate”, “estimate”, “believe”, “intend”, “plan” or “project” or the negative of these words or other variations on these words or comparable terminology. Forward-looking statements are subject to a number of risks and uncertainties, many of which are beyond the Company’s ability to control or predict, that may cause the actual results of the Company to differ materially from those discussed in the forward-looking statements. Factors that could cause actual results or events to differ materially from current expectations include, among other things, without limitation, the inability of the Company, through its wholly-owned subsidiary, GrowPros MMP Inc., to obtain a license for the production of medical marijuana; failure to obtain sufficient financing to execute the Company’s business plan; competition; regulation and anticipated and unanticipated costs and delays, the success of the Company’s research strategies, the applicability of the discoveries made therein, the successful and timely completion and uncertainties related to the regulatory process, the timing of clinical trials, the timing and outcomes of regulatory or intellectual property decisions and other risks disclosed in the Company’s public disclosure record on file with the relevant securities regulatory authorities. Although the Company has attempted to identify important factors that could cause actual results or events to differ materially from those described in forward-looking statements, there may be other factors that cause results or events not to be as anticipated, estimated or intended. Readers should not place undue reliance on forward-looking statements. While no definitive documentation has yet been signed by the parties and there is no certainty that such documentation will be signed The forward-looking statements included in this news release are made as of the date of this news release and the Company does not undertake an obligation to publicly update such forward-looking statements to reflect new information, subsequent events or otherwise unless required by applicable securities legislation.
Tetra Bio-Pharma Inc.
Robert (Bob) Béchard
Vice President Finance and Business Development
bob@tetrabiopharma.com
M: +1 514 817-2514
For media information, please contact:
Daniel Granger
Daniel.granger@acjcommunication.com
ACJ Communication
O: +1 514 840 7990
M: +1 514 232 1556
- Published in Tetra Bio Pharma
Tetra Bio-Pharma Inc. Announces the Closing of $4,292,000 Non-Brokered Private Placement
Momentum Public Relations
Press Release: March 28 2018
Tetra Bio-Pharma Inc.(TBP:V), a global leader in cannabinoid-based drug development and discovery, is pleased to announce it has closed its non-brokered private placement of 4,292,000 units at a price of $1.00 per unit for aggregate gross proceeds of $4,292,000. Each unit consists of one common share and one non-transferable warrant, with a whole warrant entitling the holder to purchase one common share at a price of $1.30 per share for a period of thirty-six months expiring March 28th, 2021.
The Corporation intends to use the net proceeds of the Offering to advance its Phase 3 trial for PPP001 and other clinical trials, to effect the repayment of indebtedness, for the support of its commercial efforts and for general corporate and working capital purposes.
The securities issued pursuant to the private placement will be subject to a four-month hold period from the closing date. Completion of the private placement remains subject to the receipt of all necessary regulatory approvals, including the approval of the TSX Venture Exchange.
The securities described herein have not been, and will not be, registered under the U.S. Securities Act or any state securities laws, and accordingly, may not be offered or sold within the United States except in compliance with the registration requirements of the U.S. Securities Act and applicable state securities requirements or pursuant to exemptions therefrom. This press release shall not constitute an offer to sell or the solicitation of an offer to buy securities in the United States, nor shall there be any sale of these securities in any jurisdiction in which such offer, solicitation or sale would be unlawful.
The Corporation paid commissions in association with the financing of an aggregate of $275,800 in cash and the issuance of 275,800 finders’ warrants to GMP Richardson and IA Securities, exercisable at a price of $1.00 for a period of 24 months from closing. Each finders’ warrant is exercisable into a common share and common share purchase warrant, which is exercisable at a price of $1.30 per common share purchase warrant for a period of 24 months from the closing of the financing.
The TSX Venture Exchange Inc. has not approved the contents of this press release. Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
About Tetra Bio-Pharma:
Tetra Bio-Pharma (TSX VENTURE:TBP)(OTCQB:TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and clinical development. Tetra is focusing on three core business pillars: clinical research, pharmaceutical promotion and retail commercialization of cannabinoid-based products.
More information at: www.tetrabiopharma.com
Source: Tetra Bio-Pharma
- Published in Tetra Bio Pharma
Tetra Bio-Pharma Signs Co-development and Distribution Partnership Term Sheet
Momentum Public Relations
Press Release: March 8 2018
Tetra Bio-Pharma Inc. (“Tetra” or the “Company”) (TSX VENTURE:TBP)(OTCQB:TBPMF), a global leader in cannabinoid-based drug development and discovery, is pleased to announce it has signed a co-development and distribution partnership term sheet with a major specialty healthcare distributor.
The finalization of the term sheet follows Tetra’s September 20th, 2017 Letter of Intent for a co-development and distribution partnership with the same privately held company in the specialty healthcare market. The company has a pan-Canadian distribution network that reaches up to 7000 specialized healthcare professionals. Through their distribution channel, they have access to more than 3000 specialized healthcare businesses in Canada and already work with a worldwide network of partners.
Partnership highlights
- In order to maximize both marketing and revenue impact, Tetra and its partner have agreed to launch all products at the same time, once the regulatory framework has been established by Health Canada following the legalization of cannabis. This includes cannabinoid-based as well as non-cannabinoid-based products. Both companies are confident that this strategy will solidify the brand awareness of Tetra’s partner in this new product line. The timing of the original LOI was for the commencement of revenue in Q1 2018. Tetra and its partner are now revising that date to Q4 2018.
- Both Tetra and its partner will agree to a clinical trial plan, with Tetra owning and maintaining the right to use the data gathered from the trial(s) for its own regulatory and commercial efforts in its own, non-competitive Fields of Use.
- Tetra will develop a line of private label products for its partner, the products will be distributed under the partner’s brand.
Management Statement
“We are very pleased to sign this term sheet as it solidifies the elements of the LOI outlined in September. We are continually striving and working hard every day to both solidify and grow Tetra as a major player in cannabinoid pharma and this is yet again a step in that direction. I am excited to continue to build on the momentum that we have going for us now and throughout 2018,” says Bernard Fortier, CEO of Tetra.
About Tetra Bio-Pharma:
Tetra Bio-Pharma (TSX VENTURE:TBP)(OTCQB:TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and clinical development. Tetra is focusing on three core business pillars: clinical research, pharmaceutical promotion and retail commercialization of cannabinoid-based products.
More information at: www.tetrabiopharma.com
Source: Tetra Bio-Pharma
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-looking statements
Some statements in this release may contain forward-looking information. All statements, other than of historical fact, that address activities, events or developments that the Company believes, expects or anticipates will or may occur in the future (including, without limitation, statements regarding potential acquisitions and financings) are forward-looking statements. Forward-looking statements are generally identifiable by use of the words “may”, “will”, “should”, “continue”, “expect”, “anticipate”, “estimate”, “believe”, “intend”, “plan” or “project” or the negative of these words or other variations on these words or comparable terminology. Forward-looking statements are subject to a number of risks and uncertainties, many of which are beyond the Company’s ability to control or predict, that may cause the actual results of the Company to differ materially from those discussed in the forward-looking statements. Factors that could cause actual results or events to differ materially from current expectations include, among other things, without limitation, the inability of the Company, through its wholly-owned subsidiary, GrowPros MMP Inc., to obtain a license for the production of medical marijuana; failure to obtain sufficient financing to execute the Company’s business plan; competition; regulation and anticipated and unanticipated costs and delays, the success of the Company’s research strategies, the applicability of the discoveries made therein, the successful and timely completion and uncertainties related to the regulatory process, the timing of clinical trials, the timing and outcomes of regulatory or intellectual property decisions and other risks disclosed in the Company’s public disclosure record on file with the relevant securities regulatory authorities. Although the Company has attempted to identify important factors that could cause actual results or events to differ materially from those described in forward-looking statements, there may be other factors that cause results or events not to be as anticipated, estimated or intended. Readers should not place undue reliance on forward-looking statements. While no definitive documentation has yet been signed by the parties and there is no certainty that such documentation will be signed The forward-looking statements included in this news release are made as of the date of this news release and the Company does not undertake an obligation to publicly update such forward-looking statements to reflect new information, subsequent events or otherwise unless required by applicable securities legislation.
Tetra Bio-Pharma Inc.
Dr. Anne-Sophie Courtois, DVM
Vice President, Marketing & Communications
(438) 899-7575
For investors information, please contact:
investors@tetrabiopharma.com
(438) 504-5784
- Published in Tetra Bio Pharma
Tetra Bio-Pharma names Aphria CFO as Chairman of Audit Committee
Momentum Public Relations
Press Release: February 26, 2018
Tetra Bio-Pharma Inc. (“Tetra” or the “Corporation”) (TSX VENTURE:TBP)(OTCQB:TBPMF), announced changes to its Board of Directors (the “Board”) today, including the appointment of Aphria Inc.’s Chief Financial Officer (CFO), Mr. Carl Merton, as Chair of Tetra’s Audit Committee.
In addition to his extensive financial background, Mr. Merton is a Chartered Accountant and has served as a past Chair of both the CICBV and the International Association of Professional Business Valuators. Mr. Merton is currently a member of the Board of Directors and Chair of the Audit Committee of Motor City Community Credit Union.
Mr. Merton has served on the Tetra Board since 2016 and brings over 20 years of financial and business experience to the Audit Committee. He replaces Mr. Robert Brouillette, whose resignation was accepted immediately upon the Board becoming aware that a decision had been rendered by a professional regulatory organization against Mr. Brouillette in relation to professional activities unrelated to his responsibilities as a director of Tetra.
Mr. André Rancourt, Tetra’s Chair, was also named to the Audit Committee. Mr. Rancourt also acts as a consultant on several commercial strategy committees including FIA and IRZC. He has significant practical experience that provided him with expertise in many fields, including human and animal natural health products. Over the last ten years, he worked as a consultant to re-organize the operations of companies on behalf of several venture capital investment funds.
“These appointments further strengthen our corporate governance practices,” said Tetra CEO Bernard Fortier. “Both Mr. Merton and Mr. Rancourt have stellar reputations in the capital market and will continue to provide our management team and our entire Board with support and guidance as we embark on the next stage of our growth.”
About Tetra Bio-Pharma:
Tetra Bio-Pharma (TSX VENTURE:TBP)(OTCQB:TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and clinical development. Tetra is focusing on three core business pillars: clinical research, pharmaceutical promotion and retail commercialization of cannabinoid-based products.
More information at: www.tetrabiopharma.com
Source: Tetra Bio-Pharma
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-looking statements
Some statements in this release may contain forward-looking information. All statements, other than of historical fact, that address activities, events or developments that the Corporation believes, expects or anticipates will or may occur in the future (including, without limitation, statements regarding potential acquisitions and financings) are forward-looking statements. Forward-looking statements are generally identifiable by use of the words “may”, “will”, “should”, “continue”, “expect”, “anticipate”, “estimate”, “believe”, “intend”, “plan” or “project” or the negative of these words or other variations on these words or comparable terminology. Forward-looking statements are subject to a number of risks and uncertainties, many of which are beyond the Corporation’s ability to control or predict, that may cause the actual results of the Corporation to differ materially from those discussed in the forward-looking statements. Factors that could cause actual results or events to differ materially from current expectations include, among other things, without limitation, failure to obtain sufficient financing to execute the Corporation’s business plan; competition; regulation and anticipated and unanticipated costs and delays, the success of the Corporation’s research strategies, the applicability of the discoveries made therein, the successful and timely completion and uncertainties related to the regulatory process, the timing of clinical trials, the timing and outcomes of regulatory or intellectual property decisions and other risks disclosed in the Corporation’s public disclosure record on file with the relevant securities regulatory authorities. Although the Corporation has attempted to identify important factors that could cause actual results or events to differ materially from those described in forward-looking statements, there may be other factors that cause results or events not to be as anticipated, estimated or intended. Readers should not place undue reliance on forward-looking statements. While no definitive documentation has yet been signed by the parties and there is no certainty that such documentation will be signed The forward-looking statements included in this news release are made as of the date of this news release and the Corporation does not undertake an obligation to publicly update such forward-looking statements to reflect new information, subsequent events or otherwise unless required by applicable securities legislation.
Tetra Bio-Pharma Inc.
Andre Rancourt
Executive Chairman
andre.r@tetrabiopharma.com
(438) 899-7575
- Published in Tetra Bio Pharma, Uncategorized
Tetra Bio-Pharma Announces the Completion of GrowPros Transaction
Momentum Public Relations
Press Release: February 22, 2018
Tetra Bio-Pharma Inc. (“Tetra” or the “Company”) (TSX-V:TBP) (OTCQB:TBPMF), a global leader in cannabinoid-based drug development and discovery, is pleased to announce the completion of the sale of its shares of GrowPros MMP Inc. (“GrowPros”) to North Bud Farms Inc. (the “Purchaser” or “North Bud”) as previously announced in Tetra’s December 21st, 2017 press release.
Transaction Details
As previously disclosed, Tetra will receive total proceeds in the amount of $350,000, including an initial $175,000 that was paid in December 2017, as well as 15,000,000 common shares of the Purchaser, representing approximately 41% of the Purchaser’s currently issued and outstanding share capital. It remains Tetra’s intention to have such shares be transferred to its shareholders as a dividend in kind on a pro-rata basis, concurrently with the proposed initial public offering of the Purchaser and listing of the shares of the Purchaser on a recognized Canadian stock exchange. The transaction is structured as a sale with a resolutory condition whereby, in the event that the initial public offering of the Purchaser is not completed prior to June 30, 2018 (or such other date as may be agreed upon by the Corporation and the Purchaser), the sale of the shares of GrowPros will be deemed to have never occurred and title to the shares of GrowPros will revert back to Tetra, and in turn GrowPros would then remain a wholly-owned subsidiary of Tetra.
As part of the transaction, North Bud has agreed to advance GrowPros’ late stage ACMPR application (submitted in November 2014) through the application process with Health Canada. In addition, the Purchaser intends to begin the phase 1 build out of a 50,000-square foot GMP grade indoor production facility in late Q1 2018. The facility is located on 130 acres of agricultural land in Venosta, Quebec (40 min north of Ottawa). The Purchaser believes that the property can be developed into a total of 1.5 million square feet of production space. The parties intend to enter into an agreement shortly so that Tetra will be granted access to future licenced cannabis production by GrowPros, securing another source of pharmaceutical GMP quality cannabis.
The cash proceeds will be used to further strengthen Tetra’s balance sheet and, most importantly, allow the Company to focus its activities on drug development and clinical trials of its pharmaceutical business. Tetra expects this transaction to generate long-term shareholder value by ensuring another supply of cannabis with pharmaceutical GMP quality for the pipeline of products under development.
Management Statement
“This is an important transaction for Tetra and our shareholders. Not only have we monetized an asset that brings immediate cash into the business but we also have an equity stake in North Bud that we intend to distribute to our shareholders. Of course, long term, securing an additional steady supply of dried cannabis is hugely beneficial for our pharmaceutical development programs. Finally, I am very excited that North Bud will be pushing forward the ACMPR application with Health Canada. This is a great day for all parties involved!” Says Bernard Fortier, CEO of Tetra.
About North Bud Farms:
North Bud is a partially owned subsidiary of North Bud Capital Holdings Ltd. North Bud intends to apply for a listing on a recognized Canadian stock exchange in Q2 2018. North Bud, in addition to its newly-acquired ownership of GrowPros, will be focused on GMP standardized pharma grade cannabis production as well as food grade cannabinoid infused inputs for both the international pharmaceutical market and the pending consumer goods and consumables segment of the recreational cannabis market.
About Tetra Bio-Pharma:
Tetra Bio-Pharma (TSX-V:TBP) (OTCQB:TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and clinical development. Tetra is focusing on three core business pillars: clinical research, pharmaceutical promotion and retail commercialization of cannabinoid-based products.
More information at: www.tetrabiopharma.com
Source: Tetra Bio-Pharma
For further information, please contact Tetra Bio-Pharma Inc.
Dr. Anne-Sophie Courtois, DVM
Vice President, Marketing & Communications
anne-sophie.courtois@tetrabiopharma.com
1 (438) 899-7575
- Published in Tetra Bio Pharma
New Molecules in Cannabis Discovered That Stimulate the Immune System to Destroy Tumor Cells
Momentum Public Relations
Blog: February 21, 2018
Medical Cannabis Going From Strength to Strength with New
Discovery by Pascal BioSciences
Scientific Proof of Efficacy Just Around the Corner as Canadian
Companies lead the Way
Global Cancer Drug Market expected to hit US$161. Billion in
2021
On February 21, 2018 the news broke that Vancouver-based Pascal
Biosciences, (TSXV: PAS) had identified molecules in cannabis
that stimulate the immune system to destroy tumor cells. It is the
first time that cannabinoids have been shown to activate an
immune system response.
The discovery is important because it works hand in hand with the
leading new class of cancer fighting agents known as checkpoint
inhibitors that activate the immune system to destroy cancer cells.
Using cannabinoids to enhance the recognition of cancer cells may
very well prove to enhance the efficacy of checkpoint inhibitors.
Checkpoint inhibitors are recently approved therapies that activate
the immune system to kill cancer cells. The three which are
currently on the market, Opdivo, Keytruda and Yervoy had over
US$ 6 billion in sales during 2017. Research is ongoing to develop
drugs that enhance checkpoint inhibitors. Pascal is the first to do
so.
Cannabinoids are the chemical compounds in the cannabis plant
with medical properties. More than a 100 different cannabinoid
compounds have been identified. While medical cannabis has been
available in Canada and the United States for some time its
availability and use has been based on anecdotal evidence.
Canada may very well become an international leader in medical
cannabis drug development. Tetra Bio-Pharma, (TSXV: TBP), for
instance is now conducting phase three clinical trials to prove the
safety and efficacy of a cannabinoid-based chronic and cancer pain
treatment, PPP001.
Zion Market Research predicted in 2016 that the global market for
cancer drugs would reach US$161.30 Billion by the end of 2021.
Because there has not yet been any scientific evidence that
cannabinoid-based drugs are effective or safe cannabinoid-based
treatments are in limbo. Once their safety and efficacy have been
proved they will be eligible to receive Health Canada, and in the
United States, FDA approval. This in turn will give them a Drug
Identification Number, (DIN), which in turn will allow them to be
prescribed by physicians and make them eligible for insurance
coverage.
Thanks to its phase three clinical trial to prove the safety and
efficacy of its smokable cancer and chronic pain treatment,
PPP001, it looks as if Tetra Bio-Pharma will be the first
biopharmaceutical to do so. Tetra also has a deep pipeline of
cannabinoid-based treatments for ailments such as PTSD,
anorexia, nausea and ocular pain. Tetra expects PPP001 to be on
the market as early as 2019 or at the latest during 2020.
Pascal has not yet started clinical trials.
- Published in Blog
Sun Life Signals the Start of Mass Medical Cannabis Market
Momentum Public Relations
Blog: February 16 2018
Sun Life Signals the Start of Mass Medical Cannabis Market
Aphria Becomes SAQ Recreational Marijuana Supplier
Tetra Bio-Pharma Receives Approval for Phase Three Medical Cannabis Cancer Chronic Pain Treatment
As the countdown to marijuana legalization accelerates the acceptance of medical cannabis has become more common. A defining moment has taken place with the decision by the Sun Life Assurance Company of Canada to include medical cannabis in group benefit health plans. It is the first Canadian insurance company to do so.
According to the Globe And Mail, employers will have the option of including medical cannabis coverage under an extended group health care benefit plan, starting on March 1, 2018.
As any Canadian knows Sun Life is a huge company. Its benefit plan serves 22,000 companies and covers more than five million employees and dependents.
In the Globe And Mail Story by Clare O’Hara, the senior vice-president of group benefits at Sun Life, Doug Jones, is reported to have said that the company decided to include medical cannabis coverage in its group benefit health plans because the companies it offered coverage to were asking about cannabis coverage on an increasing basis.
Employers will now have the choice about whether to include medical cannabis coverage and up to what level. Under the new system, coverage will be available from $1,500 to $6,000. So far coverage is going to be extended to five severe conditions including chronic cancer pain, nausea associated with chemotherapy and palliative care.
Eight Capital has predicted the international medical cannabis market at C$180 Billion within 10-15 Years.
Sun Life obviously sees medical cannabis coverage as a valuable commodity either in terms of customer retention or customer acquisition.
A significant factor in all of this is that Sun Life is offering coverage without the benefit of a DIN number. DIN stands for drug identification number. A DIN number is awarded to a drug that has successfully gone through clinical testing and been approved by Health Canada in Canada or the Food and Drug Administration in the United States.
Once a drug has a DIN number it can be legally prescribed by doctors and covered under insurance plans. While Canada has been allowing the sale of medical marijuana through licensed growers nobody yet has actually proved that medical marijuana works.
That is about to change. Montreal-based Tetra Bio-Pharma, (TSXV: TBP) (OTCQB: TBPMF) has just received Health Canada approval for the phase 3 clinical trial of a smokable dried cannabis prescription drug. The drug, now known as PPP001, targets chronic pain, as well as pain associated with advanced cancer.
The clinical testing is designed to prove the safety and efficacy of medical cannabis and specifically of PPP001. Tetra Bio-Pharma CEO Bernard Fortier describes it like this: “It will be a landmark trial. It will be a landmark approval. It will be the first smokable cannabis drug that will be approved, as a legitimate drug, to be prescribed by physicians.”
Tetra has a strong development pipeline featuring drugs that treat chronic pain, nausea, insomnia, PTSD, and eye ailments. As CEO Bernard Fortier says, “We are not a one molecule company.”
In late 2017 Tetra launched its first product, the trademarked Rx Princeps, a unique blend of dried medical marijuana used in its ongoing clinical trials for PPP001, a chronic pain treatment for terminal cancer patients. Rx Princeps is available for registered medical marijuana users in Canada through Tetra’s partner and licensed medical cannabis producer Aphria Inc.
Aphria, (TSXV: APH) (USOTC:APHQF) has recently been snapping up other licensed growers in the industry and is Canada’s lowest cost cannabis producer. It has also just signed a deal to become one of the six licensed growers chosen to provide marijuana to Quebec’s Societe des alcohols du Quebec (SAQ), the provincial liquor agency chosen to also distribute marijuana when legalization comes into effect. Aphria will be providing up to 12,000 Kgs of marijuana to Quebec annually.
With six different growers all offering different types of marijuana it would seem fair that Quebec is going to treat marijuana much as it does wine by providing a wide variety of product. Marijuana has come a long way but it still offers an investment opportunity in either producers or drug development companies like Tetra that may not come again.
- Published in Blog, Medical Marijuana, News Home, Tetra Bio Pharma
