InMed Begins Preclinical Work on Orofacial Pain
2015-03-18 – News Release – InMed Pharmaceuticals Inc. (IN:CSE) has initiated a program to identify and evaluate cannabinoid compounds for the treatment of chronic orofacial pain. Initial drug discovery and preclinical development are currently under way in collaboration with members of the faculty of pharmaceutical sciences at the University of British Columbia. The work is being funded by a grant from Mitacs, a leading Canadian organization that accelerates innovative projects through strategic academic and industry collaboration.
Dr. Sazzad Hossain, chief scientific officer, stated: “Pain in the orofacial region is one of the more complex and difficult-to-treat conditions for patients and clinicians. Current treatments are limited in terms of efficacy and are fraught with considerable side effects. Recent advances in cannabinoid pharmacology have renewed hope for these patients with the exciting prospect of safer, more effective cannabis-based treatments. Under this collaboration, Inmed will investigate the role of cannabinoid receptors in chronic orofacial pain, including neuropathic pain, muscle pain and arthritis of the jaw joint.”
A Mitacs grant was awarded to Dr. Hayes Wong, a PhD-level researcher, with extensive experience in developing orofacial models of pain. Dr. Wong will be working with Prof. Brian Cairns, a specialist in research for chronic pain above the neck. In conjunction with InMed, the Mitacs grant will be utilized to screen selected InMed compounds in orofacial pain models.
About Mitacs
Mitacs is a national, private, not-for-profit organization that develops the next generation of innovators with vital scientific and business skills through a suite of unique research and training programs, such as Mitacs Accelerate, Elevate, Step, Enterprise and Globalink. In partnership with companies, government and universities, Mitacs is supporting a new economy using Canada’s most valuable resource — its people.
We seek Safe Harbor.
- Published in Blog, Life Sciences, Medical Marijuana
InMed Forms Strategic Collaboration to Develop PhytoCannabinoid Therapies
2015-03-10 – News Release – InMed Pharmaceuticals Inc. (IN:CSE) has formed an exclusive strategic collaboration with the University of Debrecen, Hungary, to develop novel phytocannabinoid-based therapies to treat ocular allergic symptoms. The collaboration will leverage InMed’s proprietary intelligent cannabinoid drug design platform (IDP) and will be led by one of the world’s leading cannabinoid researchers, Dr. Tamas Biro, MD, PhD, DSc. Dr. Biro has extensive research experience in studying the endocannabinoid system (ECS) and the closely related transient receptor potential (TRP) channels in various human diseases.
Under the discovery and development collaboration InMed’s IDP platform will be used to identify cannabinoid-based and non-cannabinoid-based phytochemicals for ocular therapies focused on reducing various pro-inflammatory cytokines in in vitro and in vivo models.
Dr. Sazzad Hossain, chief scientific officer of InMed, stated: “We have accumulated significant experience and expertise in developing cannabinoids to treat ocular disease, which forms the basis of this strategic collaboration. As we prepare to initiate phase 1 clinical trials of our lead phytocannabinoid-based drug candidate CTI-085 for glaucoma, we look forward to expanding our ophthalmic therapy pipeline by developing ocular anti-allergic drugs, where we expect Dr. Biro’s 18 years of experience in this specialty field to be invaluable.”
“The global ocular allergy space is a multibillion-dollar market, and our platform offers a novel approach to potentially treating a wide array of allergies,” said Craig Schneider, president and chief executive officer of InMed. “We are excited to move a lead candidate toward preclinical and clinical trials and will provide additional updates as they become available.”
We seek Safe Harbor.
- Published in Blog, Life Sciences, Medical Marijuana
InMed Pharmaceuticals to Present Live Investor Webcast March 5
Company invites individual and institutional investors as well as advisors to attend interactive real-time presentation at VirtualInvestorConferences.com
Craig Schneider, Chief Executive Officer, will provide a company overview during a live webcast at VirtualInvestorConferences.com.
DATE: Thursday, March 5, 2014
TIME: 1:15pm ET
LINK: http://VirtualInvestorConferences.com > click the red “register/ watch event now” button
This will be a live, interactive online event where investors are invited to ask the company questions in real-time – both in the presentation hall as well as InMed’s “virtual trade booth.” If attendees are not able to join the event live on the day of the conference, an on-demand archive will be available for 90 days.
It is recommended that investors pre-register to save time and receive event updates.
About InMed
InMed is a clinical stage biopharmaceutical company that specializes in developing novel therapies through the research and development into the extensive pharmacology of cannabinoids coupled with innovative drug delivery systems. InMed’s proprietary platform technology, product pipeline and accelerated development pathway are the fundamental value drivers of the company. For more information, visit www.inmedpharma.com.
About VirtualInvestorConferences.com
Since 2010, VirtualInvestorConferences.com, created by BetterInvesting (NAIC) and PR Newswire, has been the only monthly virtual investor conference series that provides an interactive forum for presenting companies to meet directly with investors using a graphically-enhanced online platform.
Designed to replicate the look and feel of location-based investor conferences, Virtual Investor Conferences unites PR Newswire’s leading-edge online conferencing and investor communications capabilities with BetterInvesting’s extensive retail investor audience network.
- Published in Blog
InMed Pharmaceuticals on the Rise
By Frehiwote Negash –
As the marijuana debate continues to dominate headlines on both sides of the border, there are emerging players that have taken the pharmaceutical approach by developing therapeutic options using cannabinoids and testing their effectiveness via clinical trials. As marijuana gains traction as a viable medical option, it is not surprising to see the pharmaceutical companies like Otsuka, Novartis, Eli Lilly and GW Pharma all getting involved in therapy development using cannabinoids in what could be a lucrative opportunity.
In light of changing public perception, Vancouver based InMed Pharmaceuticals (C: IN, OTCQB: IMLFF) formally known as Cannabis Technologies has been on a tear in recent weeks with their share price nearly tripling. After reaching a low of $0.11 in December, the company has rebranded and teamed up with Tiberend Strategic Advisors to increase their exposure to the American market, particularly in the biotech world and is creating buzz. The price was bolstered by their recent showing at the J.P Morgan Biotech show in January and their latest announcement of a new therapy for Epidermolysis bullosa simplex; a genetic skin condition that makes the epidermis fragile and causes it to blister. This foray into developing dermatological therapies makes sense if you consider that there are no FDA approved drugs on the market for this condition. This is the third therapy in the companies’ roster of treatments with therapies for glaucoma and arthritic joint pain already in the pipeline.
While the development of new cannabinoid-based therapies is vital to the company’s fortunes, what distinguishes InMed from their competition is their unique proprietary platform technology. The platform is designed to accelerate the process in which drugs go through the requisite clinical trials and receive government approval for public consumption. Instead of the standard 10-15 year wait for FDA approval, the platform allows for development and commercialization within 3-5 years. If time is of the utmost importance, then InMed has struck gold with this platform.
Investor interest is high and for good reason. With world renowned Dr. Sazzad Hossien, Ph.D., as their Chief Scientific Officer and the recent additions of Dr. Tarek Mansour, Ph.D., a veteran in the pharmaceutical sector, and Kevin Puil, a reputable investment manager on the Board of Directors, InMed has a brilliant management team with extensive knowledge in the pharmaceutical industry and the knowhow to capitalize on a growing market. InMed will be holding an Interactive Investor Webcast on March 5th at 1:15 PM EST. This is a golden opportunity for investors to look at a company with tons of potential and a promising future.
Source: InMed Pharmaceuticals , CNN Money
- Published in Blog, Life Sciences, Medical Marijuana
InMed Pharmaceuticals (IN:CSE) Appoints Dr. Abo Mohammed as Chief Medical Officer
Inmed Pharmaceuticals announced today that it has appointed former Associate Medical Director of GW Pharmaceuticals, Abo Mohammed –
VANCOUVER, British Columbia, March 4, 2015 /PRNewswire/ — InMed Pharmaceuticals, Inc. (“InMed”) (CSE: IN; OTCQB: IMLFF), a clinical stage biopharmaceutical company specializing in the development of cannabinoid based therapies, today announced that it has appointed former Associate Medical Director of GW Pharmaceuticals, Abo Mohammed, MD, DPM, MFPM, as Chief Medical Officer, effective immediately. Dr. Mohammed is a proven leader in the development of cannabinoid therapies, having played a strategic role in the clinical development, R&D, and commercialization of these specialty drugs.
As Chief Medical Officer, Dr. Mohammed will join InMed’s executive management team and will be directly involved in developing the clinical trial strategy for InMed’s lead candidates and product development strategy for the Company’s pipeline of cannabinoid based therapies, including its two clinical stage programs, CTI-085 and CTI-091, and the recently initiated program for Epidermolysis bullosa simplex (EBS), INM-750.
“Our recent progress and anticipated advancement of our lead drug candidates have made the creation of the Chief Medical Officer position a corporate priority,” commented Craig Schneider, Chief Executive Officer of InMed Pharmaceuticals. “There are very few individuals world-wide who have Dr. Mohammed’s level of expertise and direct experience in cannabinoid drug development. We are very fortunate to have been able to attract such a qualified executive to the InMed team.”
Dr. Sazzad Hossain, Chief Scientific Officer, added, “Dr. Mohammed’s expertise in regulatory affairs, clinical development and marketing in the cannabinoid based therapies is expected to add immediate value to InMed as we seek to successfully advance our cannabinoid based therapies along the clinical and regulatory pathway.”
Dr. Mohammed commented, “Joining InMed at this transformative stage, as the company prepares for clinical development of its lead programs, is the opportunity I’d been seeking to continue my passion for bringing cannabinoid therapies to the market. I look forward to leveraging my expertise in support of InMed’s exciting drug candidates.”
Prior to joining InMed Pharmaceuticals, Dr. Mohammed served as Associate Medical Director at GW Pharmaceuticals; a UK-based Pharmaceutical Company specializing in the development of cannabinoid based prescription medicines. In this role, and others at GW Pharmaceuticals, Dr. Mohammed was involved in the advanced delivery of core clinical research and was involved in key decision-making regarding R&D and product commercialization. He played a leading role in GW Pharmaceuticals’ pharmacovigilance team where his responsibilities included handling of the company’s drug safety data from both clinical trials and post-marketing sources, and general management of safety signals including investigations, reviews and reporting. He is also a consultant to the Nigerian regulatory authority (NAFDAC) in the areas of pharmacovigilance, post-marketing surveillance and clinical trials.
Prior to joining GW Pharmaceuticals Dr. Mohammed was with PPD, a leading global contract research organization providing drug discovery, development, lifecycle management and laboratory services. Dr. Mohammed also served as Chief Medical Officer/Clinical Director in various public healthcare establishments in Africa from 1996 to November 2010.
Dr. Mohammed received his MD at Ahmadu Bello University, Zaria Nigeria followed by an MSc in Orthopaedics at University College London. Dr. Mohammed achieved a DipPharMed in Pharmaceutical Medicine at University of Wales in Cardiff. He is Member, Faculty of Pharmaceutical Medicine (Royal College of Physicians of England), the British Association of Pharmaceutical Physicians and the International Society for Pharmacovigilance.
About InMed
InMed is a clinical stage biopharmaceutical company that specializes in developing novel therapies through the research and development into the extensive pharmacology of cannabinoids coupled with innovative drug delivery systems. InMed’s proprietary platform technology, product pipeline and accelerated development pathway are the fundamental value drivers of the company.
For more information, visit www.inmedpharma.com
- Published in Blog, Life Sciences, Medical Marijuana
Cannabis Oil Cures Infant of Cancer, Dissolves Inoperable Tumor
Guest Writer for Wake Up World
According to Dr. William Courtney, the western medical mind has a very hard if not impossible time trying to understand the diverse actions of Cannabidiol.
A Doctor of Medicine with extensive education and experience in microbiology, psychiatry and forensic medicine, Dr. William Courtney explains how his 8 months old patient had a massive centrally-located, inoperable brain tumor. The child’s father pushed for “non-traditional” treatment utilizing cannabis and put cannabinoid oil on the baby’s pacifier twice per day, gradually increasing the dose. Within two months there was a dramatic reduction in the size of the baby’s tumor.
Dr. Courtney pointed out that the success of the cannabis approach means that “this child… is not going to have the long-term side effects that would come from a very high dose of chemotherapy or radiation”.
Cannabis Cures Inoperable Tumor
While 10,000 year old cultural practices involve drying then heating cannabis to effect a nearly complete decarboxylation of THC-Acid into THC, the creation of massive amounts of THC is compounded by the introduction of a psychoactive side effect that has a 10 mg dose limitation secondary to CB1 receptor stimulation.
Research conducted in Bethesda Maryland led to Patent 6,630,507 held by the United States of America since 2003 that teaches that the lack of psycho-activity in CBD allows doses that are 100-200 times greater than the tolerable dose of THC. The articulated “effective oral human dosage schedule is 20 mg / kg body weight” requires a considerable amount of cannabis. The simplest approach is to consume the trichrome-laden fully-mature flower along with the 80-day leaf. Patient responses have exceeded any expectations.
Historically, dietary use of the entire raw cannabis plant brings us back in line with 34 million years of cannabis evolution. Lipid messenger molecules preceded cannabis by billions of years. The 4 billion year old development of lipid messenger molecules not only regulated resource management in the most primitive life forms, but were central in the earliest autocrine and paracrine modulation of cellular function. I believe autocrine cross talk was the necessary precedent to symbiotic, then multi-cellular life forms. Tissue specific or paracrine cluster regulation is the domain of the lipid messenger molecules and is the path to comprehending the incredible diversity of function that are only now beginning to be understood. While our perception / publication of these physiologic properties are new, the phenomenal beneficial affects were there yesterday, last year, if not hundreds of millions to billions of years ago.
Researchers have now found that the compound, called cannabidiol, has the ability to ‘switch off’ the gene responsible for metastasis in an aggressive form of cancer. Importantly, this substance does not produce the psychoactive properties of the cannabis plant.
Cannabinoids can prevent cancer, reduce heart attacks by 66{92d3d6fd85a76c012ea375328005e518e768e12ace6b1722b71965c2a02ea7ce} and insulin dependent diabetes by 58{92d3d6fd85a76c012ea375328005e518e768e12ace6b1722b71965c2a02ea7ce}. Dr. Courtney recommends drinking 4 – 8 ounces of raw flower and leaf juice from any Hemp plant, 5 mg of Cannabidiol (CBD) per kg of body weight, a salad of Hemp seed sprouts and 50 mg of THC taken in 5 daily doses.
Learn more: 5 Diseases Proven To Respond Better To Cannabis Than Prescription Drugs
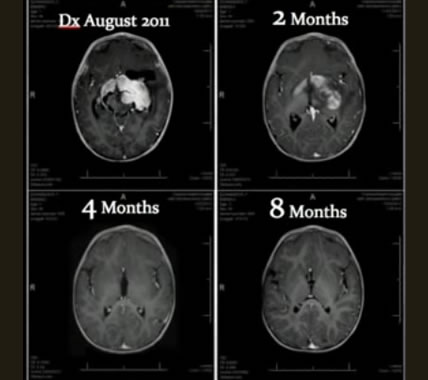
The progression of healing in the infant as the tumor (large white center mass in upper left scan) gradually disappeared in 8 months through treatment with only cannabinoids.
“If you heat the plant, you will decarboxylate THC-acid and you will get high, you”ll get your 10 mg. If you don’t heat it, you can go up to five or six hundred milligrams & use it as a Dietary Cannabis… and push it up to the Anti-oxidant and Neuro-protective levels which come into play at hundreds of milligrams”, stated Dr. William Courtney.
“The Hemp plant is actually an excellent plant because the THC content can be low, that’s if you’re treating a condition for which appears CBD food supplement is in order… The plants we’re using in Luxembourg have only 1{92d3d6fd85a76c012ea375328005e518e768e12ace6b1722b71965c2a02ea7ce} CBD, a 1{92d3d6fd85a76c012ea375328005e518e768e12ace6b1722b71965c2a02ea7ce} CBD plant is providing you with 19 times more CBD per pound than Oranges provide you of vitamin C. A 1{92d3d6fd85a76c012ea375328005e518e768e12ace6b1722b71965c2a02ea7ce} is an excellent source, you can make tremendous concentrates you can eat the plant raw – and the absence of the THC – you can heat Hemp, which you can’t do with other strains because the THC acid comes out and you end up with a psycho-toxic substance”, says Dr. Courtney.
27th July 2014
By Marco Torres
- Published in Medical Marijuana
If Big Pharma Isn’t Nervous, It Should Be; Early Stage Cannabis Technologies…Potentially the Next GW Pharma
VANCOUVER, British Columbia, July 31, 2014 (GLOBE NEWSWIRE) — Deep in the brain, buried within the central nervous system as well as lymphatic tissues and organs throughout the body are cannabinoid receptors; patiently waiting to help address a myriad of diseases. With the advent of cannabis research and therapy development many patients will likely trigger them very soon.
Two known receptors in the Endocannabinoid system are CB1 and CB2. There is mounting evidence that there are many more. Simply put, this system of receptors is involved in dealing with a variety of physiological processes including appetite, pain-sensation, mood and memory.
Activating these receptors by introducing the appropriate drug based on a specific formula of cannabis, primarily utilizing Cannabidiol (CBD) and Tetrahydrocannabinol (THC) has already shown remarkable potential efficacy, albeit somewhat anecdotal, in the treatment of a host of afflictions ranging from cancer to epilepsy, glaucoma, MS, Tourette’s and even eczema.
To date there have been approximately 100 cannabinoids identified; each with the potential to be an integral component of a lower cost treatment; countering the expensive and frequently toxic Big Pharma drugs and therapies.
The current level of research and development of cannabis therapies is analogous to where the Internet was in the mid 1990’s. What is known is that the introduction of targeted phytocannabinoid formulations, such as those with CBD and THC, signal the body to make more endocannabinoids and open more cannabinoid receptors enhancing the body’s ability to fight pain and disease.
“At this point, we don’t actually know how many therapies are possible utilizing phytocompounds, but we suspect hundreds, if not thousands,” stated Craig Schneider, President and CEO of Cannabis Technologies (CAN: CSE, CANLF: OTCQB). “To that end, CAN has developed a proprietary Cannabinoid Drug Design Platform (CDP) to identify new bioactive compounds within the cannabis plant that interact with certain genes responsible for specific diseases.”
The poster stock in this Life Sciences sector is GW Pharmaceutical. The Company IPO’d at $8.90 in May 2013 and traded as high as $107 in 2014. When investors compare the metrics of peers GWPH and CAN, the case for the latter appears compelling.
While it would be easy to draw the usual David and Goliath analogy, in this case the participants, while competitors are really more peers, working toward the same therapeutic goals. And, as a result of the focused CDP development process, cannabis therapies can be on the market in 4-6 years versus 10-15 years as is the norm through the Big Pharma pathway.
GW currently trades at $87 has a market cap of $1.5 billion and had trailing twelve-month (ttm) revenues of $50 million, is virtually debt free and has approximately $163 million in cash. Cannabis Technologies trades at $0.37 is pre-revenue and has a market cap of $12 million with roughly $600k in cash. CAN shares have a 2014 high of $0.71 and low of $0.33.
GWPH market cap is 30 times revenue. Translating that multiple to eventual revenues to early stage CAN evidences compelling growth potential.
GWPH, as CAN, decided early on to dedicate R&D to therapy development and plant their respective flags firmly in Life Science space instead of the class of ‘Medical Marijuana’ companies with all the different connotations.
These companies are involved in serious and life saving science. There are others as well, including AbbVie, which makes the FDA approved chemotherapy nausea treatment Marinol, which is a synthetic formulation of THC. Valeant Pharmaceuticals produces Cesamet, which is a like treatment. The best known to investors is likely GW’s vapor delivered Sativex, used currently in 25 countries outside the US for treatment of the spasticity associated with MS. Sativex is currently in clinical trials for approval as a treatment of cancer pain.
CAN’s Schneider notes: “The media has categorized CAN as an early-stage GW Pharma, a comparison we welcome. We are currently entering Phase 1 trials for our glaucoma treatment CTI-085, which showed great therapeutic promise in pre-clinical trials relieving the ocular pressure associated. This initial therapy is much more, being a proof of concept of the ability of our CDP to identify specifically engineered treatments to deal with many debilitating and deadly diseases.”
For context, the $12 billion ocular disease market includes $5.7 billion for glaucoma.
Other drugs in development include GW’s Epidolex for the treatment of rare diseases as well as other compounds in clinical trials for treatment of autoimmune, diabetes and schizophrenia.
The key to therapy going forward is this specific engineering and the ability to replicate the compound for quality and consistency. Sativex is basically 50{92d3d6fd85a76c012ea375328005e518e768e12ace6b1722b71965c2a02ea7ce} CBD and 50{92d3d6fd85a76c012ea375328005e518e768e12ace6b1722b71965c2a02ea7ce} THC. The CBD component has the dual task of being the active ingredient as well as damping down the psychotropic effects of the THC. CBD comes from the hemp plant and has only trace THC.
As new cannabis drugs develop, individual formulations will be more therapy specific, have non-cannabis ingredients added and undergo stringent quality and consistency controls.
Big Pharma has a right to be nervous. Side effects from cannabis therapies are virtually non-existent, development costs are extremely low by comparison–$5 billion on average per Big Pharma drug—and companies like CAN are confident that as it progresses it can develop therapies in a period of 60-90 days instead of decades.
Part of the strategic engineering is not just how much of this and that goes into a compound. The key is to develop plants that produce the right material for each formulation. It is not inconceivable that if there were 500 cannabis therapies, there would be 500 different strains of cannabis plant as ‘feed stock’.
The label of Medical Marijuana companies, when referring to enterprises such as GW, CAN, AbbVie and Valeant, are the exception to what appears to be a wild west show at times. These are serious life science companies. To include them with the plethora of Medical Marijuana initiatives, whether junior mining companies looking for a new direction or those that feel simply growing generic marijuana is a sound business plan, many will likely fail or be swallowed.
Like the Internet of old where there are few survivors today from that era, the cannabis space will eventually be littered with casualties as it builds out. What is not in dispute, is that the efficacy of cannabis appears undeniable and therapy development will continue and likely speed up, building on early successes. GW and CAN will likely be among those to grow and prosper; a good thing for both shareholders and, more importantly, those millions of patients suffering from particularly nasty diseases and conditions.
Innovative science requires scientists. Rounding out CEO Craig Schneider’s 20 years of capital market and biopharmaceutical experience CAN has two world-class scientists. Dr. Sazzad Hossain Ph.D, M.Sc., Chief Science Officer, brings two decades of experience in new drug discovery and natural health product development. His practical experience includes senior scientist at the NRCC bringing and has generated over $500 million in revenue from therapies he has been involved in developing from the discovery to commercialization.
Key as well is the Company’s breeding, genetics and cultivation division led by Dr. Hyder Khoja, Ph.D., M.Sc., A.Ag. who brings 17 years of extensive research and business provenance in life sciences and business services.
Management has spoken frequently about addressing larger therapy markets including cancer, metabolic diseases and pain and inflammation. On par with GW, CAN has plans to produce medicines in-house initiated by its CDP technology, breeding and cultivation division and proprietary formula engineering. Even at this early stage, the Company is keenly aware of the need for not just the development of therapies but the ability to replicate each with strict quality and consistency.
Further adding to shareholder value is a patent pending for CAN’s CDP and a plan to protect IP by filing patents as therapies are developed.
If you are considering investing in the cannabis space, buy the science. Hype has a very short shelf life.
Legal Disclaimer/Disclosure: A fee has been paid for the production and distribution of this Report. This document is not and should not be construed as an offer to sell or the solicitation of an offer to purchase or subscribe for any investment. No information in this article should be construed as individualized investment advice. A licensed financial advisor should be consulted prior to making any investment decision. Financial Press makes no guarantee, representation or warranty and accepts no responsibility or liability as to its accuracy or completeness. Expressions of opinion are those of the authors only and are subject to change without notice. Financial Press assumes no warranty, liability or guarantee for the current relevance, correctness or completeness of any information provided within this article and will not be held liable for the consequence of reliance upon any opinion or statement contained herein or any omission. Furthermore, we assume no liability for any direct or indirect loss or damage or, in particular, for lost profit, which you may incur as a result of the use and existence of the information, provided within this article.
Also, please note that republishing of this article in its entirety is permitted as long as attribution and a back link to FinancialPress.com are provided. Thank you.
CONTACT: Cannabis Technologies
#350-409 Granville Street, Vancouver, BC
Canada, V6C 1T2
Tel: 604.669.7207
Fax: 604.683.2506
info@cannabis-tech.com
- Published in Life Sciences, Medical Marijuana
46 Reasons why Cannabis Technologies (CAN) will Succeed
Cannabis Technologies has begun trading as InMed Pharmaceuticals (C.IN) since the writing of this article.
1) Investing in biotech can be extremely challenging. There is always a chance that someone will come out with a better product. Typically, it takes a decade or more to approval, followed by commercialization. It usuallycosts tens of millions, to billions of dollars per drug. This can be highly dilutive to a small startup.
2) What if one company had a proprietary cookie cutter system targeting a dozen diseases quicker, cheaper and more effective? I am not sure what Cannabis Technologies will be called in a year from now. I think it will have a new name… perhaps GW Pharmaceuticals or Eli Lily? Or Novartis?
3) Dr. Hossain was chiefly responsible for a $157 million deal with Novartis a decade ago. There was also a $376 million deal with Teva in 2012 based on one of his discoveries. More on that later.
4) Dr. Tarek Mansour (Pfizer) was responsible for multiple FDA approved drugs, where the market value exceeded $1 billion. (Zeffix, Troxatyl, Bosulif, Neratinib and PFE384)
5) The big question is...how many therapy product launches, and strategic partnerships will it take before Big Pharma catches on?
6) The amplitude of possibilities dictates that Cannabis Technologies will one day be a dominant player in the prescription cannabinoid medicine market.
7) Many people now know there are at least 85 different cannabinoids isolated from cannabis exhibiting various effects that could prove therapeutic.
8) They also know that cannabidiol (CBD) alone has shown therapeutic benefits to at least 16 diseases.
9) CAN will take advantage of Strain differences to develop drugs for specific diseases, including:
Glaucoma, Inflammation/Pain/Arthritis, Huntington’s, Epilepsy, Diabetes, Obesity, Cancer & Angiogenesis
10) CAN’s proprietary Cannabinoid Drug Design Platform (“CDP”) allows computer science, statistics, mathematics and engineering to study biological data and processes from the cannabis plant which can be targeted to develop therapies for specific diseases and conditions.
11) What will CAN’s CDP be worth to a company that wants to compete with GWPH…or a better question..what would it be worth to GWPH?
12) The main active ingredients in this are the cannabinoids THC and CBD, but other pharmacologically active cannabinoids are also present and are being investigated.
13) This Platform Technology, combined with CAN’s world renowned scientific team, will enable the company to discover therapies based on proven genomics and unique chemical fingerprints that specific cellular processes leave behind.
14) They will do this both quickly and effectively, with very little money, by outsourcing patented products that are ready for clinical trials and allow a company like Novartis (for example) to absorb all the costs through to commercialization. CAN will retain a fair and reasonable interest that may generate substantial revenue.
Track Record
Dr. Hossain’s successful financial deals based on his drug discoveries over the last 15 years include:
15) 2004: Xenon, Novartis Enter $157M Deal For Obesity Compounds
16) 2006: Xenon and Takeda Announce $75M Agreement To Develop and Commercialize XEN401 for Pain
17) 2006: Xenon Enters Into Anemia Collaboration With Roche ($7 Million for Equity, and $44 Million From Research Funding)
18) 2012: Teva inks $376M deal on Xenon pain program
19) 2009: YM Biosciences Collaboration with the National Research Council of Canada’s Biotechnology Research Institute (NRC-BRI) yielded novel anticancer antibodies that may be safer than similar drugs sold by Genentech/Roche. (another partnership resulted in the production of new breast cancer drug candidates.)
20) 2009: NRC-BRI granted Alethia Biotherapeutics exclusive, worldwide diagnostic rights to a peptide that specifically binds to tumor-associated clusterin in cancer patients.
Who should CAN target?
21) GW Pharmaceuticals has one the broadest clinical pipelines of any company in the marijuana industry…so will CAN.
22) Even though GW may be suffering the typical challenges that all biopharma companies have to endure, with costs far outstripping revenues, they had no problem raising $169.8 million last month.
23) Over 30 years ago, two young men made their way to Vancouver from Seattle to raise money for their software company, and were unfortunately turned down. Their names were Bill Gates and Paul Allen. Very few understood what they had.
24) I think CAN is in the same position today, however, that is changing with Chris Parry writing one of the first articles. Cancer survivor and canabis acdvocate Cheryl Shuman recently endorsed Cannabis Technologies as well.
25) If the billionaire activists want Marijuana to be legalized they most likely would want to facilitate the fast tracking of several revolutionary therapies that will indeed improve the lives of the millions of sufferers around the world.
26) George Soros has spent at least $80 million on the legalization effort since 1994.
27) The late Peter B. Lewis, channeled more than $40 million to influence local debates.
28) The two billionaires’ funding has been unmatched by anyone.
29) Other wealthy activists include: Google billionaire Paul Buchheit, Facebook forefathers Sean Parker and Dustin Moskovitz, and Men’s Wearhouse founder George Zimmer.
30) As of March 31, GWPH had 75 Institutional Holders (sooner or later, they will know about CAN)
31) Analyst Firms Making GWPH Recommendations: Bank of America, Cowen, Piper Jaffray, Leerink Swann, and Morgan Stanley.
Conclusion Check List:
32) World renowned scientific team with multidisciplinary expertise? Check.
33) CSO who has a track record that developed several drugs over 15 years, generating over $500m in revenues? Check
34) Global organizations, governments, and big pharma companies that have previously benefitted from the teams research? Check.
35) Proprietary Cannabinoid Drug Design Platform? Check.
36) Tools to isolate and identify chemical compounds both quickly and effectively? Check.
37) In-house Breeding, Genetics and Cultivation division? Check.
38) Capable of developing compounds for therapies in months rather than years? Check.
39) Target specific diseases and conditions? Check.
40) Outsource early-stage research and trials to conserve capital? Check.
41) Fast Forward through Phase I, Phase II & Phase III quickly and inexpensively compared to traditional Pharma? Check
42) Competitive edge from companies that rely on third-parties to manufacture their treatments? Check
43) Fully-integrated operations to lower costs and increase quality? Check
44) Capable of commercializing therapies in a 1/3 the time of traditional drug development? Check.
45) Anaglous company with a $1.5b market cap? Check
46) Developing medicines for:
– Glaucoma
– Pain and Inflammation
– Orphan Diseases
– Metabolic Disease (Obesity, Diabetes)
– Cancers and Metabolic Diseases? Check.
Dr. Sazzad Hossain, Ph.D., M.Sc.
Chief Scientific Officer
– 20 years of academic and industrial experience in new drug discovery, natural health product development
– Group Leader and Senior Scientist at Biotechnology Research Institute of National Research Council Canada, Government of Canada’s prime biotechnology research organization where he set up pharmacology laboratory to evaluate safety and efficacy of new drugs under development in the areas of cancer, cardiovascular and ocular diseases.
Dr. Tarek S. Mansour, Ph.D., M.Sc.
Scientific Advisor
– Dr. Mansour was responsible for transition of staff and projects to the Pfizer pipeline
-Founder and Chief Executive Officer of Sabila Biosciences LLC, New York
– Under his leadership, several compounds have progressed to various stages of clinical evaluation including FDA approvals and late stage development including:Zeffix, Troxatyl, Bosulif, Neratinib and PFE384.
Dr. Hyder A. Khoja, Ph.D., M.Sc., A.Ag.
Director of Botanical Research and Cultivation
– 17 years of extensive experience in a broad range of life sciences and business services with strong leadership combined with functional expertise and experience in general business management
– Industry experience spans from initiation, operation, and contribution towards developing several research and business proposals in the fields of Agriculture-Food Security, Food Safety & Sovereignty, Alternative energy, Renewable resources, Biofuels, Nutraceutical, Hydroponics, Agriculture & Land use management and Technology transfer
– Presented his work both in at federal government and academic institutions with authorship in over 18 peer-reviewed papers, primarily in genomics, plant physiology, and alternative energy.
–His work was also recognized and appeared in United Nations Food and Agriculture Organization (FAO) Environment and Natural Resources Management as a working paper for Algae-based Biofuels
Craig Schneider
One of Craig’s first companies that he worked for was Ultra Petroleum. The stock dropped from .95 to .45 before blasting through the stratosphere to a stunning pre split price of close to $200. Put another way, the market cap rose from around $3 million to close to $3 Billion!
Loyal investors were rewarded once again in 2006, as he was the cofounder of Magnum Uranium, which was taken over by Energy Fuels in 2009. Shareholders were once again exposed to another potential 1000{92d3d6fd85a76c012ea375328005e518e768e12ace6b1722b71965c2a02ea7ce} gain by 2011.
Read more at http://www.stockhouse.com/companies/bullboard/c.can/cannabis-technologies-inc#2FOuXY6qmBPTVqry.99
- Published in Medical Marijuana
Cannabis and Cannabinoids
Cannabinoids are a group of 21-carbon–containing terpenophenolic compounds produced uniquely by Cannabis species (e.g., Cannabis sativa L.) .[1,2] These plant-derived compounds may be referred to as phytocannabinoids. Although delta-9-tetrahydrocannabinol (THC) is the primary psychoactive ingredient, other known compounds with biologic activity are cannabinol, cannabidiol (CBD), cannabichromene, cannabigerol, tetrahydrocannabivarin, and delta-8-THC. CBD, in particular, is thought to have significant analgesic and anti-inflammatory activity without the psychoactive effect (high) of delta-9-THC.
One study in mice and rats suggested that cannabinoids may have a protective effect against the development of certain types of tumors.[3] During this 2-year study, groups of mice and rats were given various doses of THC by gavage. A dose-related decrease in the incidence of hepatic adenoma tumors and hepatocellular carcinoma (HCC) was observed in the mice. Decreased incidences of benign tumors (polyps and adenomas) in other organs (mammary gland, uterus, pituitary, testis, and pancreas) were also noted in the rats. In another study, delta-9-THC, delta-8-THC, and cannabinol were found to inhibit the growth of Lewis lung adenocarcinoma cells in vitro and in vivo .[4] In addition, other tumors have been shown to be sensitive to cannabinoid-induced growth inhibition.[5–8]
Cannabinoids may cause antitumor effects by various mechanisms, including induction of cell death, inhibition of cell growth, and inhibition of tumor angiogenesis invasion and metastasis.[9–12] Two reviews summarize the molecular mechanisms of action of cannabinoids as antitumor agents.[13,14] Cannabinoids appear to kill tumor cells but do not affect their nontransformed counterparts and may even protect them from cell death. These compounds have been shown to induce apoptosis in glioma cells in culture and induce regression of glioma tumors in mice and rats. Cannabinoids protect normal glial cells of astroglial and oligodendroglial lineages from apoptosis mediated by the CB1 receptor.[15]
The effects of delta-9-THC and a synthetic agonist of the CB2 receptor were investigated in HCC.[16] Both agents reduced the viability of HCC cells in vitro and demonstrated antitumor effects in HCC subcutaneous xenografts in nude mice. The investigations documented that the anti-HCC effects are mediated by way of the CB2 receptor. Similar to findings in glioma cells, the cannabinoids were shown to trigger cell death through stimulation of an endoplasmic reticulum stress pathway that activates autophagy and promotes apoptosis. Other investigations have confirmed that CB1 and CB2 receptors may be potential targets in non-small cell lung carcinoma [17] and breast cancer.[18]
An in vitro study of the effect of CBD on programmed cell death in breast cancer cell lines found that CBD induced programmed cell death, independent of the CB1, CB2, or vanilloid receptors. CBD inhibited the survival of both estrogen receptor–positive and estrogen receptor–negative breast cancer cell lines, inducing apoptosis in a concentration-dependent manner while having little effect on nontumorigenic, mammary cells.[19]
CBD has also been demonstrated to exert a chemopreventive effect in a mouse model of colon cancer.[20] In the experimental system, azoxymethane increased premalignant and malignant lesions in the mouse colon. Animals treated with azoxymethane and CBD concurrently were protected from developing premalignant and malignant lesions. In in vitro experiments involving colorectal cancer cell lines, the investigators found that CBD protected DNA from oxidative damage, increased endocannabinoid levels, and reduced cell proliferation. In a subsequent study, the investigators found that the antiproliferative effect of CBD was counteracted by selective CB1 but not CB2 receptor antagonists, suggesting an involvement of CB1 receptors.[21]
Another investigation into the antitumor effects of CBD examined the role of intercellular adhesion molecule-1 (ICAM-1).[12] ICAM-1 expression has been reported to be negatively correlated with cancer metastasis. In lung cancer cell lines, CBD upregulated ICAM-1, leading to decreased cancer cell invasiveness.
In an in vivo model using severe combined immunodeficient mice, subcutaneous tumors were generated by inoculating the animals with cells from human non-small cell lung carcinoma cell lines.[22] Tumor growth was inhibited by 60{92d3d6fd85a76c012ea375328005e518e768e12ace6b1722b71965c2a02ea7ce} in THC-treated mice compared with vehicle-treated control mice. Tumor specimens revealed that THC had antiangiogenic and antiproliferative effects. However, research with immunocompetent murine tumor models has demonstrated immunosuppression and enhanced tumor growth in mice treated with THC.[23,24]
In addition, both plant-derived and endogenous cannabinoids have been studied for anti-inflammatory effects. A mouse study demonstrated that endogenous cannabinoid system signaling is likely to provide intrinsic protection against colonic inflammation.[25] As a result, a hypothesis that phytocannabinoids and endocannabinoids may be useful in the risk reduction and treatment of colorectal cancer has been developed.[26–29]
CBD may also enhance uptake of cytotoxic drugs into malignant cells. Activation of the transient receptor potential vanilloid type 2 (TRPV2) has been shown to inhibit proliferation of human glioblastoma multiforme cells and overcome resistance to the chemotherapy agent carmustine.[30] In an in vitro model, CBD increased TRPV2 activation and increased uptake of cytotoxic drugs, leading to apoptosis of glioma cells without affecting normal human astrocytes. This suggests that coadministration of CBD with cytotoxic agents may increase drug uptake and potentiate cell death in human glioma cells.
Many animal studies have previously demonstrated that delta-9-THC and other cannabinoids have a stimulatory effect on appetite and increase food intake. It is believed that the endogenous cannabinoid system may serve as a regulator of feeding behavior. The endogenous cannabinoid anandamide potently enhances appetite in mice.[31] Moreover, CB1 receptors in the hypothalamus may be involved in the motivational or reward aspects of eating.[32]
Understanding the mechanism of cannabinoid-induced analgesia has been increased through the study of cannabinoid receptors, endocannabinoids, and synthetic agonists and antagonists. The CB1 receptor is found in both the central nervous system (CNS) and in peripheral nerve terminals. Similar to opioid receptors, increased levels of the CB1 receptor are found in regions of the brain that regulate nociceptive processing.[33] CB2 receptors, located predominantly in peripheral tissue, exist at very low levels in the CNS. With the development of receptor-specific antagonists, additional information about the roles of the receptors and endogenous cannabinoids in the modulation of pain has been obtained.[34,35]
Cannabinoids may also contribute to pain modulation through an anti-inflammatory mechanism; a CB2 effect with cannabinoids acting on mast cell receptors to attenuate the release of inflammatory agents, such as histamine and serotonin, and on keratinocytes to enhance the release of analgesic opioids has been described.[36–38] One study reported that the efficacy of synthetic CB1- and CB2-receptor agonists were comparable with the efficacy of morphine in a murine model of tumor pain.[39]
- Adams IB, Martin BR: Cannabis: pharmacology and toxicology in animals and humans. Addiction 91 (11): 1585-614, 1996. [PUBMED Abstract]
- Grotenhermen F, Russo E, eds.: Cannabis and Cannabinoids: Pharmacology, Toxicology, and Therapeutic Potential. Binghamton, NY: The Haworth Press, 2002.
- National Toxicology Program: NTP toxicology and carcinogenesis studies of 1-trans-delta(9)-tetrahydrocannabinol (CAS No. 1972-08-3) in F344 rats and B6C3F1 mice (gavage studies). Natl Toxicol Program Tech Rep Ser 446 (): 1-317, 1996. [PUBMED Abstract]
- Bifulco M, Laezza C, Pisanti S, et al.: Cannabinoids and cancer: pros and cons of an antitumour strategy. Br J Pharmacol 148 (2): 123-35, 2006. [PUBMED Abstract]
- Sánchez C, de Ceballos ML, Gomez del Pulgar T, et al.: Inhibition of glioma growth in vivo by selective activation of the CB(2) cannabinoid receptor. Cancer Res 61 (15): 5784-9, 2001. [PUBMED Abstract]
- McKallip RJ, Lombard C, Fisher M, et al.: Targeting CB2 cannabinoid receptors as a novel therapy to treat malignant lymphoblastic disease. Blood 100 (2): 627-34, 2002. [PUBMED Abstract]
- Casanova ML, Blázquez C, Martínez-Palacio J, et al.: Inhibition of skin tumor growth and angiogenesis in vivo by activation of cannabinoid receptors. J Clin Invest 111 (1): 43-50, 2003. [PUBMED Abstract]
- Blázquez C, González-Feria L, Alvarez L, et al.: Cannabinoids inhibit the vascular endothelial growth factor pathway in gliomas. Cancer Res 64 (16): 5617-23, 2004. [PUBMED Abstract]
- Guzmán M: Cannabinoids: potential anticancer agents. Nat Rev Cancer 3 (10): 745-55, 2003. [PUBMED Abstract]
- Blázquez C, Casanova ML, Planas A, et al.: Inhibition of tumor angiogenesis by cannabinoids. FASEB J 17 (3): 529-31, 2003. [PUBMED Abstract]
- Vaccani A, Massi P, Colombo A, et al.: Cannabidiol inhibits human glioma cell migration through a cannabinoid receptor-independent mechanism. Br J Pharmacol 144 (8): 1032-6, 2005. [PUBMED Abstract]
- Ramer R, Bublitz K, Freimuth N, et al.: Cannabidiol inhibits lung cancer cell invasion and metastasis via intercellular adhesion molecule-1. FASEB J 26 (4): 1535-48, 2012. [PUBMED Abstract]
- Velasco G, Sánchez C, Guzmán M: Towards the use of cannabinoids as antitumour agents. Nat Rev Cancer 12 (6): 436-44, 2012. [PUBMED Abstract]
- Cridge BJ, Rosengren RJ: Critical appraisal of the potential use of cannabinoids in cancer management. Cancer Manag Res 5: 301-13, 2013. [PUBMED Abstract]
- Torres S, Lorente M, Rodríguez-Fornés F, et al.: A combined preclinical therapy of cannabinoids and temozolomide against glioma. Mol Cancer Ther 10 (1): 90-103, 2011. [PUBMED Abstract]
- Vara D, Salazar M, Olea-Herrero N, et al.: Anti-tumoral action of cannabinoids on hepatocellular carcinoma: role of AMPK-dependent activation of autophagy. Cell Death Differ 18 (7): 1099-111, 2011. [PUBMED Abstract]
- Preet A, Qamri Z, Nasser MW, et al.: Cannabinoid receptors, CB1 and CB2, as novel targets for inhibition of non-small cell lung cancer growth and metastasis. Cancer Prev Res (Phila) 4 (1): 65-75, 2011. [PUBMED Abstract]
- Nasser MW, Qamri Z, Deol YS, et al.: Crosstalk between chemokine receptor CXCR4 and cannabinoid receptor CB2 in modulating breast cancer growth and invasion. PLoS One 6 (9): e23901, 2011. [PUBMED Abstract]
- Shrivastava A, Kuzontkoski PM, Groopman JE, et al.: Cannabidiol induces programmed cell death in breast cancer cells by coordinating the cross-talk between apoptosis and autophagy. Mol Cancer Ther 10 (7): 1161-72, 2011. [PUBMED Abstract]
- Aviello G, Romano B, Borrelli F, et al.: Chemopreventive effect of the non-psychotropic phytocannabinoid cannabidiol on experimental colon cancer. J Mol Med (Berl) 90 (8): 925-34, 2012. [PUBMED Abstract]
- Romano B, Borrelli F, Pagano E, et al.: Inhibition of colon carcinogenesis by a standardized Cannabis sativa extract with high content of cannabidiol. Phytomedicine 21 (5): 631-9, 2014. [PUBMED Abstract]
- Preet A, Ganju RK, Groopman JE: Delta9-Tetrahydrocannabinol inhibits epithelial growth factor-induced lung cancer cell migration in vitro as well as its growth and metastasis in vivo. Oncogene 27 (3): 339-46, 2008. [PUBMED Abstract]
- Zhu LX, Sharma S, Stolina M, et al.: Delta-9-tetrahydrocannabinol inhibits antitumor immunity by a CB2 receptor-mediated, cytokine-dependent pathway. J Immunol 165 (1): 373-80, 2000. [PUBMED Abstract]
- McKallip RJ, Nagarkatti M, Nagarkatti PS: Delta-9-tetrahydrocannabinol enhances breast cancer growth and metastasis by suppression of the antitumor immune response. J Immunol 174 (6): 3281-9, 2005. [PUBMED Abstract]
- Massa F, Marsicano G, Hermann H, et al.: The endogenous cannabinoid system protects against colonic inflammation. J Clin Invest 113 (8): 1202-9, 2004. [PUBMED Abstract]
- Patsos HA, Hicks DJ, Greenhough A, et al.: Cannabinoids and cancer: potential for colorectal cancer therapy. Biochem Soc Trans 33 (Pt 4): 712-4, 2005. [PUBMED Abstract]
- Liu WM, Fowler DW, Dalgleish AG: Cannabis-derived substances in cancer therapy–an emerging anti-inflammatory role for the cannabinoids. Curr Clin Pharmacol 5 (4): 281-7, 2010. [PUBMED Abstract]
- Malfitano AM, Ciaglia E, Gangemi G, et al.: Update on the endocannabinoid system as an anticancer target. Expert Opin Ther Targets 15 (3): 297-308, 2011. [PUBMED Abstract]
- Sarfaraz S, Adhami VM, Syed DN, et al.: Cannabinoids for cancer treatment: progress and promise. Cancer Res 68 (2): 339-42, 2008. [PUBMED Abstract]
- Nabissi M, Morelli MB, Santoni M, et al.: Triggering of the TRPV2 channel by cannabidiol sensitizes glioblastoma cells to cytotoxic chemotherapeutic agents. Carcinogenesis 34 (1): 48-57, 2013. [PUBMED Abstract]
- Mechoulam R, Berry EM, Avraham Y, et al.: Endocannabinoids, feeding and suckling–from our perspective. Int J Obes (Lond) 30 (Suppl 1): S24-8, 2006. [PUBMED Abstract]
- Fride E, Bregman T, Kirkham TC: Endocannabinoids and food intake: newborn suckling and appetite regulation in adulthood. Exp Biol Med (Maywood) 230 (4): 225-34, 2005. [PUBMED Abstract]
- Walker JM, Hohmann AG, Martin WJ, et al.: The neurobiology of cannabinoid analgesia. Life Sci 65 (6-7): 665-73, 1999. [PUBMED Abstract]
- Meng ID, Manning BH, Martin WJ, et al.: An analgesia circuit activated by cannabinoids. Nature 395 (6700): 381-3, 1998. [PUBMED Abstract]
- Walker JM, Huang SM, Strangman NM, et al.: Pain modulation by release of the endogenous cannabinoid anandamide. Proc Natl Acad Sci U S A 96 (21): 12198-203, 1999. [PUBMED Abstract]
- Facci L, Dal Toso R, Romanello S, et al.: Mast cells express a peripheral cannabinoid receptor with differential sensitivity to anandamide and palmitoylethanolamide. Proc Natl Acad Sci U S A 92 (8): 3376-80, 1995. [PUBMED Abstract]
- Ibrahim MM, Porreca F, Lai J, et al.: CB2 cannabinoid receptor activation produces antinociception by stimulating peripheral release of endogenous opioids. Proc Natl Acad Sci U S A 102 (8): 3093-8, 2005. [PUBMED Abstract]
- Richardson JD, Kilo S, Hargreaves KM: Cannabinoids reduce hyperalgesia and inflammation via interaction with peripheral CB1 receptors. Pain 75 (1): 111-9, 1998. [PUBMED Abstract]
- Khasabova IA, Gielissen J, Chandiramani A, et al.: CB1 and CB2 receptor agonists promote analgesia through synergy in a murine model of tumor pain. Behav Pharmacol 22 (5-6): 607-16, 2011. [PUBMED Abstract]
- Published in Blog


