Cannara Biotech Appoints Jack Kay as New Board Director
Momentum Public Relations
Press Release: April 8, 2019
Cannara Biotech (“Cannara” or the “Company”) (CSE: LOVE FRA: 8CB), an emerging vertically integrated cannabis company focused on cultivation and cannabis-infused products, today announced the appointment of Jack Kay as a Director on Cannara’s Board, effective immediately. Mr. Kay is generally recognized as one of Canada’s leading pharmaceutical executives, having held senior positions in a number of firms, including CEO of Apotex.
“A strong and independent board is not only vital for a successful public company, it can serve as a competitive advantage in the battlefield of commerce and growth,” said Zohar Krivorot, President and CEO of Cannara Biotech. “We are thrilled to welcome Jack to Cannara, all of us are excited to learn from his insights and leadership. Jack’s track record of developing new products, launching them in Canada and elsewhere, nurturing global partnerships and spearheading a 10,000-person organization speaks for itself.”
“Like many Canadians, I’m both intrigued and excited about the prospects of cannabis to make a positive difference in the health and wellbeing of patients and non-patients alike,” said Jack Kay. “I look forward to having a more engaged role in this emerging sector, especially so with a company that has a compelling business plan and the commensurate resources to execute.”
Mr. Jack Kay, for 35 years, held various roles at Apotex Inc., including Chief Executive Officer, Chief Operating officer, President and Vice Chairman. He has 50 years’ experience in pharmaceutical management and sales. Mr. Kay has served on a number of pharmaceutical and biotech boards, including as the Chairman of Helix Biopharma Corp and Cangene Corp, and a director at Barr Pharmaceuticals Inc. He has also served in senior capacities for a number of industry and community organizations, including as the Chairman of Humber River Regional Hospital (Toronto), the International Schizophrenic Society, the Canadian Drug Manufacturers Association, the Canadian Schizophrenia Foundation and the Canadian Generic Pharmaceutical Association. Mr. Kay has degrees from both from the University of Manitoba and McGill University.
About Cannara Biotech
Cannara Biotech (CSE:LOVE) is building one of the largest indoor cannabis cultivation facilities (625,000 square feet) in Canada and the largest in Quebec. Leveraging Quebec’s low electricity costs, Cannara Biotech’s facility will produce high-grade indoor cannabis and cannabis-infused products for the Canadian and international markets.
The CSE nor its Regulation Services Provider accepts responsibility for the adequacy or accuracy of this release.
- Published in Cannabis, Cannara, Life Sciences, Marijuana, Medical Marijuana, News Home
North Bud Farms Signs Binding Letter of Intent to Enter U.S. Market with Strategic Acquisition of Multi-State Licensed Operator Eureka Vapor
Momentum Public Relations
Press Release: March 6, 2019
North Bud Farms Inc. (CSE: NBUD) (OTCQB: NOBDF) (“NORTHBUD” or the “Company”) is pleased to announce that effective March 3, 2019 it entered into a binding letter of intent (“LOI”) to acquire all the issued and outstanding shares of Eureka Vapor LLC. and all of its subsidiaries (“Eureka”), a U.S. multi-state cannabis operator, and arm’s length to the Company, in a transaction valued at CAD$20 million.
Eureka, through its wholly-owned subsidiaries holds Manufacturing and Distribution licenses in the states of California and Colorado. Eureka manufactures and sells a premium line of disposable vapor pens as well as multi-use cartridge-style vapor pens and hardware. Eureka has been operating in California and Colorado since 2011 and 2015, respectively, showing significant organic growth year over year. In 2018, Eureka recognized revenue of approximately CAD$11.5 million* with a net profit margin of 16%* from its California and Colorado operations. Eureka anticipates further growth in revenue due to anticipated changes to retail regulation of adult cannabis use in California. Eureka products are currently available in over 100 retail stores. (*all figures are unaudited). For more information about Eureka Vapor, visit: www.eurekavapor.com.
Transaction Terms
The proposed transaction (the “Transaction”) is structured as a share purchase agreement whereby in exchange for the purchase of all of the shares of Eureka, NORTHBUD will issue CAD$20 million in common shares (“Common Shares”) to the shareholders of Eureka (the “Eureka Shareholders”) with the price per Common Share to be determined based on a formula of the higher of (a) CAD$0.35 per Common Share and (b) the 30-day volume weighted average price (“VWAP”) calculated on the closing date (the “Closing Date”) of a definitive agreement in respect of the transaction (the “Definitive Agreement”). NORTHBUD and Eureka expect to enter into the Definitive Agreement by May 30, 2019. 10% of the Common Shares issued pursuant to the Definitive Agreement will be issued to the Eureka Shareholders on the Closing Date, with the remainder of Common Shares issued in equal tranches of six, twelve, eighteen, and twenty-four months from the Closing Date (the “Escrow Period”). The Transaction will be considered a “Fundamental Change” pursuant to the policies of the CSE and will accordingly require a new listing statement (the “Listing Statement”). Given that effective as of the Closing Date the Company will have United States cannabis operations, the Listing Statement will provide disclosure of the risks associated with cannabis operations in the United States. Closing of the Transaction is subject to applicable corporate and regulatory approvals as well as shareholder and CSE approval.
In addition, Eureka Shareholders will be eligible to receive up to an additional CAD$25 million of Common Shares (“Revenue Milestone Shares”) based on the achievement of USD$25 million of revenue derived from existing Eureka California and Colorado operations. Eureka Shareholders will receive Revenue Milestone Shares pro rata, on a quarterly basis, based on the percentage of USD$25 million of revenue generated in that quarter. All Revenue Milestone Shares will continue to be subject to the remainder of the Escrow Period at the time of issuance and will only be releasable in accordance with the Escrow Period. The Revenue Milestone Shares will be issued at the 10-day VWAP at the time of issuance.
“The opportunity to partner with a recognized brand in some of the most developed retail markets in North America is an exciting development for NORTHBUD,” says Ryan Brown, CEO of NORTHBUD. “We believe that vape cartridges represent a high margin and high-growth product segment of the market. The Eureka team are proven operators and possess an unmatched product knowledge which is evidenced by the strong brand loyalty that they have established.”
“Aligning ourselves with NORTHBUD provides Eureka with both exposure to the Canadian public markets as well as the largest federally legal adult-use market in the world,” says Justin Braune, CEO of Eureka Vapor. “We will be working with the NORTHBUD team to introduce our product line into the Canadian market for the fourth quarter of 2019 when vape pens will be permitted.”
Granting of Stock Options
The Company also announces the granting of 150,000 stock options to a consultant and employee. Each option entitles the holder to acquire one Common Share for a period of five years at an exercise price of CAD$0.35 per Common Share. The options all vest immediately.
About Eureka Vapor LLC.
Headquartered in Los Angeles, California, EUREKA Vapor was founded in 2011 and holds licenses in both California and Colorado. EUREKA Vapor’s multi state operation manufactures and sells a premium line of vaporizer cartridges, disposable vapor pens and proprietary vaporizer batteries designed to work with their highly sought-after CO2 extracted oil. Using their refined extraction processes and techniques developed over almost a decade of extracting, EUREKA Vapor is committed to providing the cleanest and safest natural oil cartridges in the industry. Long referred to as one of the leaders in the industry, EUREKA has one of the most loyal customer bases in the category which reflects their commitment to honesty and transparency above all else. EUREKA continually looks for innovative ways to improve and refine their product offerings in order to deliver the best, most consistent vaping experience in the industry.
For more information, visit: www.eurekavapor.com
About North Bud Farms Inc.
North Bud Farms Inc., through its wholly-owned subsidiary GrowPros MMP Inc., is pursuing a licence under The Cannabis Act. The Company is constructing a state-of-the-art purpose-built cannabis production facility located on 95 acres of Agricultural Land in Low, Quebec. North Bud Farms Inc. will be focused on Pharmaceutical and Food Grade cannabinoid production in preparation for the legalization of edibles and ingestible products scheduled for October 2019.
For more information, visit: www.northbud.com
Neither the Canadian Securities Exchange (the “CSE”) nor its Regulation Services Provider (as that term is defined in the policies of the CSE) accepts responsibility for the adequacy or accuracy of this release.
Forward-looking statements
Certain statements included in this press release constitute forward-looking information or statements (collectively, “forward-looking statements”), including those identified by the expressions “anticipate”, “believe”, “plan”, “estimate”, “expect”, “intend”, “may”, “should” and similar expressions to the extent they relate to the Company or its management. The forward-looking statements are not historical facts but reflect current expectations regarding future results or events. This press release contains forward- looking statements including those relating to the projected growth of Eureka in 2019, the entering into of the Definitive Agreement, closing of the Transaction and associated approvals, Eureka’s ability to achieve milestones under the Definitive Agreement and associated Common Share issuances, the growth of the vape industry and its profitability, the timing of the introduction of Eureka vape pens into the Canadian market, and the projected legalization of edibles and ingestible products scheduled for October 2019. These forward-looking statements are based on current expectations and various estimates, factors and assumptions and involve known and unknown risks, uncertainties and other factors. Such risks and uncertainties include, among others, the risk factors included in North Bud Farms Inc.’s final long form prospectus dated August 21, 2018 which is available under the issuer’s SEDAR profile at www.sedar.com.
FOR ADDITIONAL INFORMATION, PLEASE CONTACT:
North Bud Farms Inc.
Edward Miller
VP, IR & Communications
Office: (855) 628-3420 ext. 3
investors@northbud.com
- Published in Cannabis, Marijuana, Medical Marijuana, News Home, NorthBud
Two in Three (65%) Canadians Would Take a Drug Containing Cannabis if Prescribed by a Doctor and Covered by Insurance
Momentum Public Relations
Press Release: February 26, 2019
When It Comes to Cannabis, Trust in Doctors’ Judgement Runs High
- Majority of Canadians (54%) believe taking cannabis for medical reasons without consulting a doctor poses a risk.
- Knowledge of Canadian clinical trials investigating cannabis medicines is low.
- Slim majority of Canadians agree that their doctor is informed enough to treat them with cannabis.
- Majority of Canadians do not consider themselves as cannabis users.
OTTAWA, Feb. 26, 2019 /CNW/ – According to a recent Ipsos poll commissioned by Tetra Bio-Pharma, it seems that Canadians believe that Doctor Knows Best when it comes to recommending cannabis for a medical ailment, even though they are less convinced that they are well enough informed to treat them with it. That concern should change as cannabinoid derived prescription drugs become approved by Health Canada, available in pharmacies and covered by private and/or public drug plans. That time is soon approaching as numerous clinical trials investigating the safety and efficacy of cannabinoid-derived drugs are currently underway.
The study revealed that two in three (65%) Canadians would be willing (36% very/29% somewhat) to take a pharmaceutical drug containing cannabis that their doctor prescribed, if it was approved by Health Canada and covered by either public or private insurance. Those more likely to be willing to take these drugs include men (69%), those aged 18-34 (72%), and residents of Ontario (71%). By contrast, if these drugs were not covered by public or private insurance and patients had to pay out of pocket, four in ten (38%) would still be willing to do so (14% strongly/25% somewhat), particularly among men (44%), those aged 18-34 (51%), and residents of BC (50%).
According to Dr. Guy Chamberland, Chief Executive Officer and CSO of Tetra Bio-Pharma, a global leader in the discovery and development of cannabinoid-derived products, the Ipsos results reinforce current understanding of consumer and physician behaviors. “Patients are open to cannabis as a medical treatment but want their healthcare professional to be in charge. On the flip side, doctors, medical bodies and payors need the safety and efficacy data that they expect from any drug they prescribe. Treatment with cannabis is complex, which is why the pharmaceutical pathway assures precise dosing and consistent formulation.”
Tetra Bio-Pharma undertook the research to gain a better understanding of the attitudes, behaviours and opinions of Canadians on cannabis drugs, including their confidence in taking them, the incidence of medical conditions they have where cannabis treatment could be used, as well as any barriers to cannabis drugs.
While much attention has been paid to the recent legalization of recreational cannabis in Canada, the Ipsos study shows that 69% of respondents do not consider themselves to be cannabis users. Of those that do, half say they use it for recreational purposes. Approximately a quarter of these users report taking it exclusively for medical purposes. If that number seems low, it could be because medical doctors and medical associations are still waiting for scientific data before recommending cannabis to their patients.
New Avenues for Pharmaceutical Research
Canadians seem confident that cannabis has a therapeutic benefit and would be willing to take it for pain and associated symptoms. An overwhelming majority (82%) of Canadians agree (34% strongly agree/48% somewhat) that cannabis can reduce pain and other symptoms. Furthermore, over two-thirds (68%) of Canadians are willing (30% very/38% somewhat) to take cannabis to help manage chronic pain, insomnia, anxiety, or depression. This is potentially interesting for those who suffer from conditions that could be treated with cannabis-based medicines. Not surprisingly, this proportion reaches 84% (60% strongly/24%) among those who suffer from three or more conditions that have been linked to treatment with a cannabis-based medicine.
When it comes to expanding access for patients to cannabis medicines through the regulatory pathway, one in six Canadians (17%) say they are closely following the progress being made with Health Canada approving clinical trials for cannabis-based medicines, while just over four in ten (41%) say they have heard of this development, but are not following it at all. Another 18% said they thought cannabis medicines already existed and the remaining one-quarter (24%) said they were not at all aware.
While cannabis is being authorized for medical purposes under the Cannabis Act, the products currently available are not Health Canada approved prescription drugs. Several clinical trials are underway in Canada with a view to investigating the safety and efficacy of cannabinoid-derived pharmaceuticals. Understandably, those suffering from one or more conditions that could be treated with medical cannabis are more likely to be following this news closely. Furthermore, over four in ten (43%) report being willing (23% very/21% somewhat) to participate in a clinical trial testing cannabis-based medicines if these trials are approved by Health Canada and they were qualified for the trial.
Doctor Knows Best
Canadians seem to recognize that turning to cannabis to manage a health condition is not something they should do on their own. Almost nine in ten (88%) believe that there is some degree of risk (25% major/29% moderate/34% minor) in taking cannabis for health conditions without consulting a physician. This should be a comforting thought considering the side effects that cannabis can have for individuals suffering from mental illness, cardiac conditions and diabetes, among other conditions.
Canadians’ level of trust in their doctor remains high when it comes to their judgement over whether to prescribe cannabis. Just under three-quarters (72%) agree (29% strongly /43% somewhat) that they would trust a drug containing cannabis if their doctor prescribed it. Interestingly though, not everyone is fully confident that their doctor is up-to-date on cannabis treatments. Only a slim majority of Canadians agree that their doctor knows how to treat them with cannabis (56%; 14% strongly/42% somewhat) and that their doctor is sufficiently well-informed on how to do so (53%; 13% strongly/41% somewhat). This suggests that Canadians believe there is room for an improvement in knowledge. It is widely believed that the integration of cannabinoid-derived drugs within the Canadian medical system will become a catalyst for the education and training of doctors on how to prescribe cannabis, something that is complex given the need for precise dosing and consistent product quality.
If ordinary Canadians are open to taking cannabis-based medicines, it seems that doctors are also receptive to prescribing them to patients. While only one in ten (11%) Canadians has asked their doctor to prescribe cannabis for a health condition, 45% of those who did so report that their doctor prescribed cannabis willingly. Another 20% said their doctor prescribed cannabis, but was hesitant, while 11% reported that their doctor prescribed another drug instead.
About the Survey
These are some of the findings of an Ipsos poll conducted between December 7 and December 10, 2018, on behalf of Tetra Bio-Pharma. For this survey, a sample of 2,002 Canadians aged 18+ was interviewed online via the Ipsos I-Say panel. The poll is accurate to within ±2.5 percentage points, 19 times out of 20, had all Canadian adults been polled.
About Tetra Bio-Pharma
Tetra Bio-Pharma (TSX-V: TBP) (OTCQB: TBPMF) is a biopharmaceutical leader in cannabinoid-derived drug discovery and development with a Health Canada approved and FDA reviewed clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. Tetra Bio-Pharma has subsidiaries engaged in the development of an advanced and growing pipeline of Bio Pharmaceuticals and Natural Health containing cannabis and other medicinal plant-based elements. With patients at the core of its mission, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies. For more information visit: www.tetrabiopharma.com.
SOURCE Tetra Bio-Pharma Inc
View original content to download multimedia: http://www.newswire.ca/en/releases/archive/February2019/26/c6186.html
- Published in Life Sciences, Medical Marijuana, News Home, Tetra Bio Pharma
Grown Rogue Announces Binding Agreement for Michigan Cannabis Licenses and Assets
Momentum Public Relations
Press Release: February 25, 2019
Grown Rogue International Inc. (CSE:GRIN | OTC: NVSIF) (“Grown Rogue” or the “Company“), a vertically-integrated, multi-state cannabis company, with licenses and operations in Oregon and California, has signed a binding agreement which provides the Company the right to acquire operational control of certain cannabis licenses and related assets as part of its expansion into the Michigan cannabis market, pending certain regulatory approvals.
“With the second highest total number of medical cannabis card holders in the United StatesMichigan’slegalization of cannabis for adult-use presents a very large cannabis market opportunity. Significant barriers to entry at the local level add meaningful value to the limited number of municipal licenses approved,” explained Obie Strickler, CEO of Grown Rogue. “We are very diligent in our expansion strategy and in finding the right partners to take the proven Grown Rogue platform into the state. Combining our expertise enables us to efficiently navigate and comply with the regional regulatory environment and rapidly expand the Grown Rogue footprint and brand.”
Grown Rogue has entered into a binding agreement (the “Agreement“) with Blue Zebra Community LLC (“Blue Zebra“). The Agreement provides for the option to acquire operational control (the “Option“) of the following cannabis related assets in Michigan pending Municipal and State regulatory approval (the “Michigan Assets“):
- Two strategically located proposed provisioning centers (retail dispensaries) in high demand regions in Midtown Detroit and Hazel Park where limited municipal licenses have been granted.
- A proposed 19,000 sq ft indoor cultivation and processing facility in Detroit, Michigan capable of producing 1,500,000 grams of dried cannabis flower annually at full production; and
- An entity that has received multiple municipal cultivation licenses for a 28-acre parcel located in the northern portion of the lower Michigan peninsula.
Once fully licensed, the provisioning (retail) centers will offer multiple Michigan cannabis brands as well as locally produced Grown Rogue branded products for the Michigan cannabis market. This will launch the Grown Rogue brand into the Midwest region.
Upon exercising the Option by Grown Rogue, Blue Zebra (or affiliates) is required to assign its rights and obligations to the Company pursuant to a binding agreement Blue Zebra (or affiliates) has with Helios Holdings, LLC (“Helios“) which provides the framework for the acquisition of the Michigan Assets as described below (the “Helios Agreement“). As part of its Agreement with Blue Zebra, Grown Rogue will issue 2,212,876 common share purchase warrants to Blue Zebra (or its affiliates) with an exercise price of $0.44 per share (the “Warrants“), which vest according to certain milestones in accordance with the agreement with Blue Zebra.
The Warrants expire on June 20, 2023. Grown Rogue will have the right to accelerate the expiry date of 25% of the Warrants during the term if the shares of the Company close at or above $1.00 per share for a period of twenty (20) consecutive days. An additional 25% of the Warrants will accelerate if the shares of the Company close at or above $1.50 per share for a period of twenty (20) consecutive days, and the remainder of the Warrants will accelerate if the shares of the Company close at or above $2.00 per share for a period of twenty (20) consecutive days.
Pursuant to the terms of the Agreement, Grown Rogue has granted Blue Zebra, together with any affiliates, a pre-emptive right to maintain ownership, should the Warrants be exercised, of up to 5% of Grown Rogue’s common shares. At the time Blue Zebra, or any affiliates, exercises its Warrants to obtain 5% ownership in the Company, Blue Zebra will have the right to nominate one member to Grown Rogue’s board who shall be nominated by management at each annual shareholder meeting of Grown Rogue until such time the Blue Zebra’s ownership in Grown Rogue falls below 4.67%. In addition, Grown Rogue has agreed to pay Blue Zebra between 5% and 7% of top line future revenues generated from its licensed operations in Michigan. Payment on these revenues shall be in a combination of stock and cash.
Helios Agreement
Helios, and its affiliate, intend to contribute real property for a 19,000 sq ft. proposed cultivation and processing facility to a newly formed joint venture to be operated by Grown Rogue, or its designated affiliate, upon receiving all necessary regulatory approvals.
Grown Rogue intends to secure a non-dilutive, real estate financing facility to cause the improvements to the Michigan Assets to be completed.
Helios will contribute the remaining Michigan Assets into one or more newly formed operating company(s) (“OpCo“). It is intended that Grown Rogue will hold a 3.62% convertible debenture for the initial funding of OpCo, with a put/call option to acquire all the issued and outstanding shares of the OpCo. The convertible debenture principal, including any interest, shall be returned to Grown Rogue first from 50% of any cash flow generated by OpCo. Conversion of the debenture by Grown Rogue is subject to State and Municipal regulatory approval.
The State licensing and regulatory process in Michigan requires multiple tiers of approval for any operations (cultivation, retail). Several municipal licenses have successfully been awarded to Helios and its affiliates.
Once licensed by the State, the 19,000 square foot facility will be the first cultivation and manufacturing center for OpCo. Initial plans for this facility will include the ability to produce 1,500,000 grams of cannabis flower per year with the construction of a perpetual harvest facility expected to open in Q4 2019. This facility will also include best-in-class extraction facilities where the OpCo will produce branded derivative products.
The interest in the 28-acre cultivation facility can include either indoor or greenhouse operations which are currently being evaluated. Construction and operation of this facility is expected in 2019 with an anticipated yearly yield of between 1,500,000 and 2,500,000 grams of annual cannabis production based on final design plans.
Both of OpCo’s proposed retail dispensaries are located in desirable high traffic locations and are expected to be licensed and operational by Q4 2019. OpCo aims to further expand to 10 retail dispensaries and 50,000 sq ft of cultivation facilities by the middle of 2020.
“Our partnership with Grown Rogue has accelerated our operations and already we have identified several additional licenses which could make Grown Rogue one of the leading cannabis operations in the entire state of Michigan,” stated Maxim Ermakov, Helios Executive Director.
About Grown Rogue
Grown Rogue International (CSE: GRIN | OTC: NVSIF) is a vertically-integrated, multi-state cannabis company curating innovative products to provide consumers with the right cannabis experience. Each of Grown Rogue’s products and strains are categorized and marketed based on unique effects and designed for the full range of a consumers’ lifestyle. Grown Rogue is scaling the vertically integrated model into multiple states by incorporating best-in-class manufacturing facilities and a proprietary distribution platform based on Microsoft technology. Grown Rogue’s diverse cannabis product suite includes premium flower, patent-pending nitrogen sealed pre-rolls, oil and concentrates, and edibles featuring a partnership with world-renowned chocolatier, Jeff Shepherd.
Subscribe to Grown Rogue investor news alerts.
- Published in Cannabis, Grown Rogue, Marijuana, Medical Marijuana, News Home
Tetra Bio-Pharma unit launches Hemp Energy Drink
Momentum Public Relations
Press Release: February 19, 2019
Tetra Natural Health, a subsidiary of Tetra Bio-Pharma Inc., has made its Hemp Energy Drink available in 59 grocery stores and convenience stores in British Columbia, Alberta and Saskatchewan.
Hemp Energy Drink tasting in Kelowna on Feb. 3, 2019
In British Columbia the Hemp Energy Drink is available to consumers in Vancouver, Balfour, Burnaby, Castlegar, Clearwater, Grand Forks, Hope, Houston, Kaslo, Keremeos, Ladner, Langley, Lillooet, Nanaimo, Nelson, North Vancouver, Oliver, Osoyoos, Port Alberni, Robson, Rossland, Salt Spring Island, Simon Fraser University, Slocan Park, Squamish, Summerland, Surrey, Trail, Vernon, Victoria, West Kelowna, Whistler and White Rock.
In Alberta, the Hemp Energy Drink can be found in Athabasca, Banff, Boyle and Langdon, and in Saskatchewan in Kindersley and Tisdale.
The tastings of the Hemp Energy Drink were very popular on Feb. 2 and Feb. 3, during the Wellness Show Vancouver 2019 and at a product presentation in Kelowna. The distribution plan is expected to accelerate the penetration of HED attractive green cans in stores in the coming weeks.
“Our distribution plan is on schedule and the arrival of the Hemp Energy Drink is creating a buzz among consumers as soon as the product becomes available in store,” said Richard Giguere, chief executive officer, Tetra Natural Health. “We will accelerate the deployment of our market opening strategy in a few weeks with the arrival of 1.7 million cans of Hemp Energy Drink, of which more than 600,000 will be quickly shipped to new markets in Canada.”
Hemp Energy Drink is the first energy drink made from hemp in the country. Light, energizing, fresh, Hemp Energy Drink is licensed by Health Canada and contains fewer calories and more natural ingredients than any other energy drink. The product will soon be available in three flavours, classic, mango and raspberry, and it will be accessible in stores in a few months in all regions across Canada.
About Tetra Bio-Pharma Inc.
Tetra Bio-Pharma is a biopharmaceutical leader in cannabinoid-based drug discovery and development with a Health Canada-approved, and Food and Drug Administration-reviewed, clinical program aimed at bringing novel prescription drugs and treatments to patients and their health care providers. The company has several subsidiaries engaged in the development of an advanced and growing pipeline of biopharmaceuticals, natural health and veterinary products containing cannabis and other medicinal plant-based elements. With patients at the core of what it does, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies.
- Published in Business, hemp, Marijuana, Medical Marijuana, News Home, Tetra Bio Pharma
Cannara Biotech – www.shopCBD.com
Cannara Biotech (CSE:LOVE)(FRA:8CB)
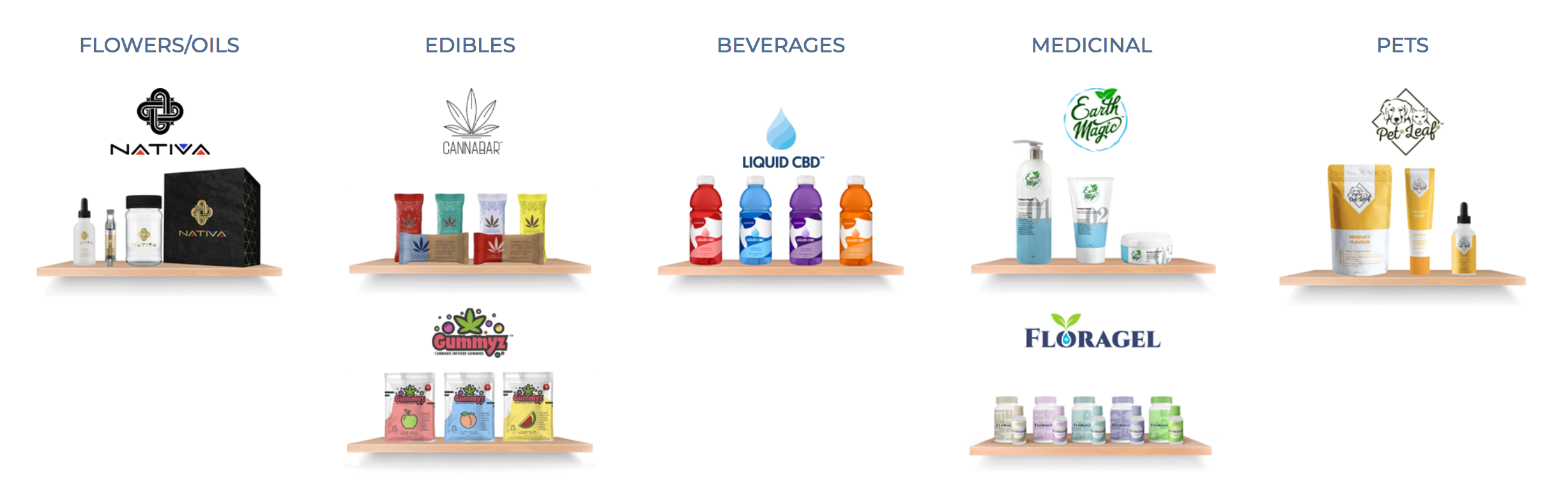
Headquartered in Montreal, Cannara Biotech is entering the U.S. THC-CBD market, through a subsidiary, with an on-line e-commerce platform called shopCBD.
“We are taking a leadership role by creating a hub for vendors to showcase their products and consumers to purchase hemp-CBD products in a fast and easy way. We want to become the Amazon of CBD,” – ZOHAR KRIVOROT – CEO and Founder
Highlights
- Zohar Krivorot, president and chief executive officer of Cannara Biotech is a 15-year veteran in the tech and on-line industries.
- Cannara Biotech is building the largest indoor cannabis cultivation facility in Quebec and one of the largest in Canada
- shopcbd.com is a platform that will showcase hemp-CBD retail products including tinctures, oils, capsules, body care, vape cartridges and pet-related CBD infused treats
- The indoor facility offers lower risk of airborne infections, mold/mildew and produces a higher grade of cannabis
- The new e-commerce platform will showcase retail products from hemp-CBD manufacturers for the U.S. consumer market
- shopCBD will provide a user-friendly on-line experience where consumers can purchase, review and compare a variety of CBD products
Trending
- With the recent passing of the 2018 Farm Bill in the U.S., CBD represents an emerging sector with many vendors seeking national reach to U.S. customers.
- The company’s strategy is to offer a wide variety of products from leading CBD brands, with competitive pricing and delivery times across the U.S.
- The rise in popularity for CBD products is driven by consumers seeking natural-based health and wellness solutions to treat various ailments from inflammation, anxiety to insomnia amongst many others.
- The hemp-derived CBD market is expected to reach $22-billion (U.S.) by 2022 according to industry analysts at Brightfield Group.
Alongside the e-commerce, on-line platform, Cannara Biotech will have on-site independent THC and CBD brands, following the adoption of regulations on cannabis-infused edibles and beverages, that will produce and ship from the facility. Cannara Biotech’s facility will be a multi-purpose facility that will cultivate recreational cannabis and produce cannabinoid-infused products including edibles, cosmetics, pet products and beverages. These products will be shipped to stores and retail outlets in Canada and to international markets.
Cannara Biotech went public in Q1 of this year and is being traded on the CSE under the ticker “LOVE” and on the FRA under the ticker “8CB”. As of February 12th, 2019, LOVE closed at .22.
—
Isabelle Arsenault
Media Relations
MomentumPR
- Published in Cannara, Marijuana, Medical Marijuana
Tetra Bio-Pharma Provides Update on its Neuropathic and Chronic Pain Drug Development Program
Momentum Public Relations
Press Release: February 12, 2019
Tetra Bio-Pharma Inc. (“Tetra” or the “Corporation“) (TSX VENTURE: TBP) (OTCQB: TBPMF), a global leader in cannabinoid-derived drug development and discovery, today announced accelerated development of its second-generation drug inhalation program for fibromyalgia and uncontrolled pain over the last months.
A Phase 1 study in healthy volunteers using vaporized PPP001 was successfully completed (click here) and the Corporation filed a patent application after unexpectedly discovering the unique composition of cannabis smoke. In July 2018, the Corporation entered into a co-development agreement with Storz & Bickel, widely recognized as the global leader in vaporizer design and manufacturing. Tetra incorporated the vaporizer into its smoke-vapor research program as it aimed to understand how inhaled cannabis relieves pain for patients.
About Neuropathic Pain (Fibromyalgia) and Chronic Pain
Fibromyalgia affects about 4 million U.S. adults or about 2% of the adult populationi. The main symptom associated with the condition is the wide range of pain symptoms that can be deep, sharp, dull, throbbing, or aching, affecting the muscles, tendons, and ligaments around the joints throughout the entire body. Treatments aimed at pain relief are the primary treatment intervention including an increasing use of opioids. Chronic pain is also associated with numerous other medical conditions including rheumatoid arthritis, osteoarthritis, gastro intestinal problems, cancer, surgery, HIV/AIDS. The global fibromyalgia treatment market was valued at approximately $10 billion dollars in 2017ii.
Tetra gained knowledge and understanding as to how smoked and vaporized cannabis works to relieve pain, based on its double-blind randomized placebo-controlled studies of PPP001 administered with the titanium pipe and Mighty Medic vaporizer and the use of smoke-vapor trapping studies to quantify exposure to the respiratory tract. A significant finding of the study was learning how much of each cannabinoid and terpene is delivered to the human body as the result of smoking or vaporizing a dried cannabis product. The key to the success of a smoked cannabis product is closely linked to its speed of delivery to the brain. In addition, the inhalation mode of delivery results in significantly higher maximal plasma levels at the peak of treatment. What does this mean? Essentially, results from our study in humans showed that the peak therapeutic levels show only a 1-minute difference in delivery between the pipe and vaporizer. Combined with the speed, the levels of cannabinoids delivered at this peak of exposure are what results in the benefits seen in patients who suffer from pain.
The pipe or vaporizer delivered important differences in some cannabinoids and terpenes. Tetra is using this insight to accelerate the development of alternative formulations for the treatment of fibromyalgia and uncontrolled chronic pain. This knowledge is incorporated in the filed patent application and based on this, Tetra will use isolates or synthetic-based supplies of cannabinoids and terpenes to develop these prescription cannabinoid-derived medicines.
During the last quarter of 2018, the Corporation began preparations for a research site in the United States to conduct its fibromyalgia clinical trial and we are intent on expanding clinical sites in Canada and in the USA in 2019.
“We previously communicated that after reviewing the human clinical data, Tetra anticipated proceeding into phase 2 clinical trials in fibromyalgia patients,” said Dr. Guy Chamberland, CEO and CSO of Tetra Bio-Pharma. “We are committed to this target and plan to accelerate this development in the USA as well. Tetra has and continues to gain tremendous credibility within the medical community. We earned this credibility because of our clinical studies and exhaustive characterization of the pharmaceutical composition of cannabis-derived smoke and vaporization products. Overall, 2018 was a productive year after completing several clinical trials and we expect 2019 to build on this as we accelerate this program.”
About Tetra Bio-Pharma
Tetra Bio-Pharma (TSX-V: TBP) (OTCQB: TBPMF) is a biopharmaceutical leader in cannabinoid-based drug discovery and development with a Health Canada approved, and FDA reviewed, clinical program aimed at bringing novel prescription drugs and treatments to patients and their healthcare providers. The Company has several subsidiaries engaged in the development of an advanced and growing pipeline of Bio Pharmaceuticals, Natural Health and Veterinary Products containing cannabis and other medicinal plant-based elements. With patients at the core of what we do, Tetra Bio-Pharma is focused on providing rigorous scientific validation and safety data required for inclusion into the existing bio pharma industry by regulators, physicians and insurance companies.
For more information visit: www.tetrabiopharma.com
- Published in Business, Medical Marijuana, News Home, Tetra Bio Pharma
Cannara Biotech Shares to Begin Trading on the Frankfurt Exchange
Momentum Public Relations
Press Release: February 11, 2019
Cannara Biotech Inc. (“Cannara Biotech” or the “Company”) (CSE: LOVE) (FRA: 8CB), a vertically integrated cannabis company focused on cultivation and cannabis-infused products, today announced its common shares are listed for trading on the Frankfurt Stock Exchange under the trading symbol “8CB”. The Company’s common shares continue to be listed on the Canadian Securities Exchange under the trading symbol “LOVE”.
The Frankfurt Stock Exchange is the tenth largest stock exchange in the world by market capitalization and third largest in terms of volume. It is Germany’s largest stock exchange.
“No doubt the world is becoming keenly interested in the global cannabis industry, and much of that enthusiasm and attention is turning towards Canadian companies who are at the forefront of innovating and leading this sector,” said Zohar Krivorot, President and CEO of Cannara Biotech. “For Cannara Biotech, whose vision is to be a premium global cannabis company, this international listing will enable us to broaden our shareholder base, while allowing international investors to participate in our growth.”
The Canadian cannabis market is forecasted at $5.2B in year one of legalization and is expected growth of 20% annually to reach $10.8B in five years.
About Cannara Biotech Inc.
Cannara Biotech is building one of the largest indoor cannabis cultivation facility (625,000 square foot) in Canada and the largest in Quebec. Leveraging Quebec’s low electricity costs, Cannara Biotech’s facility will produce high-grade indoor cannabis and cannabis-infused products for the Canadian and international markets. Most recently, the Company entered the U.S. CBD-hemp market with an online eCommerce platform called shopCBD.com, which will also make the Company an aggregator of U.S. CBD-hemp products. For more information, visit our website: www.cannara.ca
The CSE does not accept responsibility for the adequacy or accuracy of this release.
Cautionary Statement Regarding “Forward-Looking” Information
This information release contains certain forward-looking information. Such information involves known and unknown risks, uncertainties and other factors that may cause actual results, performance or achievements to be materially different from those implied by statements herein, and therefore these statements should not be read as guarantees of future performance or results. All forward-looking statements are based on the Company’s current beliefs as well as assumptions made by and information currently available to it as well as other factors. Readers are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this press release. Due to risks and uncertainties, including the risks and uncertainties identified by the Company in its public securities filings, actual events may differ materially from current expectations. The Company disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
View original content:http://www.prnewswire.com/news-releases/cannara-biotech-shares-to-begin-trading-on-the-frankfurt-exchange-300792906.html
SOURCE Cannara Biotech Inc.
View original content: http://www.newswire.ca/en/releases/archive/February2019/11/c8723.html
Contact:
Sabrina Williams, Communications Manager, sabrina.williams@cannara.ca, T: 514-543-4200 ext. 265; Zohar Krivorot, President & CEO, zohar@cannara.ca; Lennie Ryer CPA, CA, CFE, Chief Financial Officer, lennie@cannara.ca
- Published in Cannabis, Cannara, Marijuana, Medical Marijuana
Cannara Biotech Enters Cannabinoid-Infused Beverage Product Category
Momentum Public Relations
Press Release: February 7, 2019
Cannara Biotech Inc. (“Cannara Biotech” or the “Company”) (CSE: LOVE), a Montreal-based company that is building a vertically integrated cannabis company focused on cultivation and cannabis-infused products, today announced it has entered into a letter of intent with a leading Quebec microbrewery to develop cannabinoid-infused beverages. Following the regulations relating to edibles and beverages coming into effect next October, consumers will be able to purchase a variety of cannabinoid-infused beverages including non-alcoholic beer, kombucha and cider.
“Many of the world’s most successful beverage companies have aggressively invested in Cannabinoid-infused product development underscoring this product category’s market potential,” said Zohar Krivorot, President and CEO of Cannara Biotech. “This agreement also underscores our commitment in building Cannara Biotech into a premium Canadian cannabis company by leveraging our facility’s unique size and flexibility, partnering with industry leaders and supporting these joint ventures with both financial and human capital.”
Cannabinoid-infused beverage sales are forecasted to reach $260 million for North America by 2022, according to Canaccord Genuity. In addition to non-alcoholic cannabinoid-infused beer, this is the first of many partnerships that we intend to use the facility to execute our collaboration strategy to produce edibles, pet-products, cosmetics and other beverages for the Canadian market.
About Cannara Biotech Inc.
Cannara Biotech is building the largest indoor cannabis cultivation facility in Quebec, a modern and secure 625,000 square foot facility located in Farnham, less than 45 minutes from Montreal. Leveraging Quebec’s low electricity costs, Cannara’s facility will produce high-grade indoor hydroponically grown cannabis and derivative products for the Canadian and international markets. Working with partners, Cannara will generate licensing revenues, rental revenues and revenue streams from joint-venture arrangements leveraging this valuable property. For more information, visit our website: www.cannara.ca
The CSE does not accept responsibility for the adequacy or accuracy of this release.
Cautionary Statement Regarding “Forward-Looking” Information
This information release contains certain forward-looking information. Such information involves known and unknown risks, uncertainties and other factors that may cause actual results, performance or achievements to be materially different from those implied by statements herein, and therefore these statements should not be read as guarantees of future performance or results. All forward-looking statements are based on the Company’s current beliefs as well as assumptions made by and information currently available to it as well as other factors. Readers are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this press release. Due to risks and uncertainties, including the risks and uncertainties identified by the Company in its public securities filings, actual events may differ materially from current expectations. The Company disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
View original content:http://www.prnewswire.com/news-releases/cannara-biotech-enters-cannabinoid-infused-beverage-product-category-300791611.html
SOURCE Cannara Biotech Inc.
View original content: http://www.newswire.ca/en/releases/archive/February2019/07/c2566.html
Contact:
Sabrina Williams, Communications Manager, sabrina.williams@cannara.ca, T: 514-543-4200 ext. 265; Zohar Krivorot, President & CEO, zohar@cannara.ca; Lennie Ryer CPA, CA, CFE, Chief Financial Officer, lennie@cannara.ca
- Published in Cannabis, Cannara, Marijuana, Medical Marijuana
